October 20, 2016 report
Self-assembled monolayers of NHCs on gold have ballbot-type motion
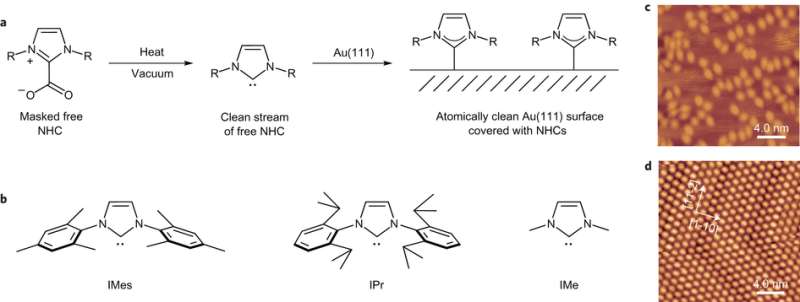
(Phys.org)—Self-assembled monolayers are molecules anchored to a surface. These molecules have been used to advance the fields of molecular sensors and electronics. Monolayers on gold surfaces are typically anchored by a thiol. One alternative to thiols are N-heterocyclic carbenes (NHCs). First isolated in 1991, NHCs have proved an important molecule for practical uses because it can serve as a ligand for transition metals as well as some main group elements. NHCs have also been used for organocatalysis, cross-coupling reactions, and pharmaceuticals.
Research groups from Westfälische Wilhelms-Universität in Münster led by Professors Harald Fuchs and Frank Glorius, used scanning-tunneling microscopy analyses combined with first principles calculations of representative NHC monolayers on gold surfaces to determine how the monolayers can be both chemically robust and highly mobile. They found that the NHC bound to gold forms an adatom that then traverses the gold surface in a ballbot-type motion. Their report appeared in a recent issue of Nature Chemistry.
NHCs form a particularly strong bond with gold because of the sp2 hybridized lone pair of electrons on the carbon atom (See figure of NHCs above). Studies have shown that NHCs can be used to form particularly stable self-assembled monolayers (SAMs) on gold surfaces and can even be modified after formation of the monolayer, but these studies are difficult to compare with each other because they do not have similar R groups.
The nitrogen-substituted R groups play a significant role in the behavior of the NHCs. Bulky R groups prohibit dimerization but may affect the stability of monolayer formation. Also because the nitrogen atoms are part of an aromatic ring, they serve as electron donors to the substituents. The type of substituent affects the electronics of the molecule.
In this study, Wang et al. used three different NHCs that were synthesized in the Glorius group, each of which had R groups that had previously been studied and represented a range of steric and electronic properties. These groups include IMes which has 2,4,6 trimethyl phenyl rings as the N-substituents; IPr which has 2,6-diisopropylphenyl substituents; and IMe which just has methyl groups as the N-substituents.
Beginning with carboxylate versions of IMes, IPr, and IMe, they deposited the NHCs onto the gold surfaces via ultrahigh vacuum vapor deposition. XPS confirmed that the NHC, not the carboxylate version, was bound to the gold surface. Scanning tunneling microscopy (STM) showed that IPr and IMe made neat, controlled surfaces, while IMes molecules were less ordered.
At various surface coverage densities, the IPr surface would form islands that broke apart and recombined. This indicated that the surface was mobile, but there was no change to morphology of the gold surface. Despite this mobility, the bond between the NHC and the gold surface proved to be highly stable at high temperatures.
This behavior may be explained by the formation of adatoms, or the interaction of one NHC molecule with one gold atom. This adatom then moves around the surface as one entity, which the authors liken to ballbot movement. They confirmed this structure by looking at the height of the IPr monolayers.
DFT calculations helped to explain how exactly these adatoms formed and moved. IPr and IMe likely bound to a gold atom that was pulled from the surface. Simultaneously, a gold atom from the second layer moves into the vacant hole. The gold atom that was pulled from the surface was bound irreversibly to the NHC to form an adatom that then readily moved across the surface.
Additional free energy calculations looked at the effects of bulky R groups, specifically comparing IPr with IMes. Wang et al. found that the pi interactions between the phenyl rings and the surface play a role in adatom formation and mobility. They reasoned that there is a balance between steric hindrance, as with IPr, and pi interactions, as displayed with IMes. And, indeed, the non-bulky IMe compound, which was more mobile than IPr and IMes, was able to form dimeric and trimeric complexes with one gold atom.
This study provides important insights into how NHC-Au monolayers form and behave. They seem to form a stronger bond than thiols with gold, but they also display mobility that is influence by the identity of the nitrogen substituents. The NHCs that tend to have weaker pi interacts with the gold surface readily forming adatoms that then move across the surface like a ballbot.
More information: Gaoqiang Wang et al, Ballbot-type motion of N-heterocyclic carbenes on gold surfaces, Nature Chemistry (2016). DOI: 10.1038/NCHEM.2622
Abstract
Recently, N-heterocyclic carbenes (NHCs) were introduced as alternative anchors for surface modifications and so offered many attractive features, which might render them superior to thiol-based systems. However, little effort has been made to investigate the self-organization process of NHCs on surfaces, an important aspect for the formation of self-assembled monolayers (SAMs), which requires molecular mobility. Based on investigations with scanning tunnelling microscopy and first-principles calculations, we provide an understanding of the microscopic mechanism behind the high mobility observed for NHCs. These NHCs extract a gold atom from the surface, which leads to the formation of an NHC–gold adatom complex that displays a high surface mobility by a ballbot-type motion. Together with their high desorption barrier this enables the formation of ordered and strongly bound SAMs. In addition, this mechanism allows a complementary surface-assisted synthesis of dimeric and hitherto unknown trimeric NHC gold complexes on the surface.
Journal information: Nature , Nature Chemistry
© 2016 Phys.org



















