How did the modern biological cell acquire its transport system?
The inside of a present day plant or animal cell quite closely resembles a busy city. Like an urban metropolis with different districts interlinked by a traffic network, a cell has distinct compartments connected to each other by a dynamic transport system.
One set of such interlinked compartments - the Golgi complex - is essential for many cellular functions, and a question that has long puzzled scientists is: how did such a complex compartment and traffic system arise within a cell?
Scientists from the National Centre for Biological Sciences have a possible answer to this question through a mathematical approach to explore how such organisation could have evolved. Somya Mani and Mukund Thattai from the Simons Centre at NCBS have shown that the Golgi complex with its attendant traffic system can emerge spontaneously from a simple model with no need for a special selection mechanism.
Within cells, the Golgi complex is a set of compartments that is essential for processing, packaging and transporting proteins and other molecules. A key characteristic of the Golgi is its organisation as a 'maturation chain' with different compartments having variable molecular compositions. These compartments perform different processing and packaging functions, especially in the synthesis and transport of giant proteins like collagen.
"Now, it's very strange that this maturation system developed in order to transport giant molecules. You can't transport large molecules if you don't have the whole system working, but if there was no function for the maturation system, what drove it's evolution?" asks Mukund Thattai. "It's a classic chicken-and-egg problem or what you call a catch-22," he says.
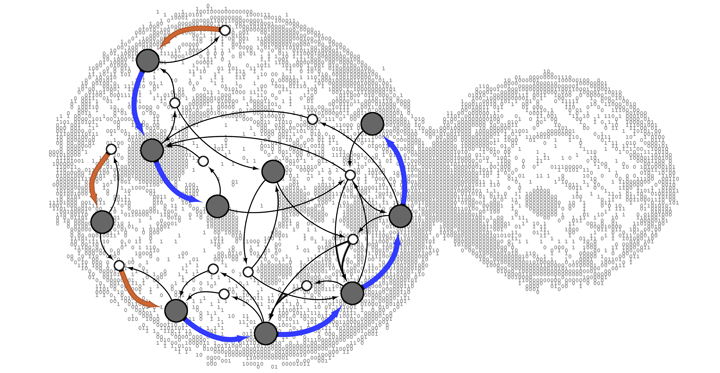
The different compartments of the Golgi complex are connected to each other and to other cellular areas via mobile membrane-bound chambers called vesicles. Vesicles constantly bud off or fuse with compartments, forming a cell-wide transport system for different types of molecules.
In order to investigate the origins of the Golgi complex, Mani and Thattai simulated this traffic system. Built on broad and simple rules, they modelled the stream of vesicles budding out of source compartments and fusing with target compartments within a cell. These events were specified by budding and fusion matrices to create a collection of simulated cellular traffic networks that had settled into a state of equilibrium.
Now, Mani and Thattai did something unconventional - they filled up the budding and fusion matrices at random. Therefore, budding and fusion events were random, with no specific purpose guiding these events.
But then, they got an astonishing result.
In roughly 25% of their simulations, the researchers came across traffic networks that had developed distinct patterns closely resembling those of a Golgi complex. This means that despite the lack of a selection mechanism for budding or fusion, a vesicular traffic network in a cell could give rise to a functional 'maturation chain' of compartments purely by chance.
In other words, Mani and Thattai's work shows that the evolution of the Golgi complex is likely to have been non-adaptive - no selection system need have pushed cells to develop a Golgi complex.
"We could argue that the Golgi might have come about for some other purpose, a purpose that nobody has been able to figure out. But the essence of this work, is that there is no other purpose," says Thattai about the work that has been described in a recent publication in the journal eLife. "But once cells had the Golgi, fine! It's a great system for transporting giant molecules, and so cells used it," he continues.
The scientists are now planning to use their model to study infectious systems like tuberculosis and HIV, which are caused by intracellular parasites that hijack a cell's vesicular traffic system for their own use.
"The parameters of our model can be easily interpreted in terms of biological properties of traffic molecules. So, our model might give us clues into the molecular mechanisms that parasites like M. tuberculosis use to hijack the vesicular transport systems," says Mani. "We can also study mechanisms that the cell could use to reinstate its normal traffic system after such an infection," she adds.
More information: DOI: 10.7554/eLife.16231.001
Journal information: eLife
Provided by National Centre for Biological Sciences