June 3, 2016 report
New biomarker for heart disease using atomic force microscopy
(Phys.org)—Researchers from the University of Lisbon and the North Lisbon Hospital Center (Centro Hospitalar Lisboa Norte) conducted a pilot study that demonstrated how atomic force microscopy (AFM) could be used as a non-invasive diagnostic tool for chronic heart disease. Their work appears in the recent issue of Nature Nanotechnology.
The World Health Organization estimates that 17.5 million people die annually due to cardiovascular diseases. Ischemic cardiomyopathy is the most common cause of heart failure and typically comes with a poor prognosis for patients. Ischemic chronic heart failure (CHF) occurs when the heart cannot receive enough blood, often due to arterial blockage or scars from heart attacks. Because CHF is so pervasive, researchers have sought biomarkers to indicate patient risk for re-occurrence, including diagnosis of ischemic versus non-ischemic CHF.
Using AFM techniques and building on their prior studies on how fibrinogen, a protein found in blood plasma, binds to the cell membrane of erythrocytes (i.e., red blood cells), Ana Filipa Guedes, Filomena A. Carvalho, Ines Malho, Nuno Lousada, Luis Sargento, and Nuno C. Santos observed molecular changes that occur in patients with ischemic CHF. These changes could be used to assess patient risk as well as provide potential targets for drug discovery. They tested fifteen patients with ischemic CHF, fifteen with non-ischemic CHF, and fifteen healthy patients as the control group.
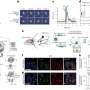
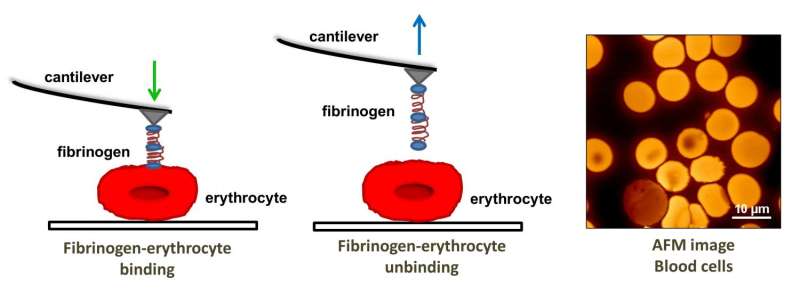
Atomic force microscopy allows for molecular-level studies because its probe tip size is on the order of nanometers in diameter. In this study AFM was used to investigate the binding strength and frequency between fibrinogen and the αvβ3 receptor on the erythrocyte surface as well as erythrocyte elasticity via tip penetration depth.
The AFM tip was functionalized with fibrinogen molecules and put in contact with the receptors on the erythrocyte surface. The binding strength was found by force measurements when the tip was pulled away from the receptor sites. Guedes, et al. found that the binding strength and frequency were different in each type of sample. Patients with ischemic CHF showed the strongest binding strength, but the lowest binding frequency. Non-ischemic CHF was next, followed by the control group, which showed the highest binding frequency but the lowest binding strength between fibrinogen and the αvβ3 receptor.
Erythrocytes are known to change shape based on their physiological environment. Erythrocytes will change their shape when a high or low shear force, or flow rate, is applied to them. This helps the red blood cells flow smoothly and not become clogged. Additionally, certain diseases can cause changes in the erythrocyte's shape. An erythrocyte's ability to change shape is based on how elastic its membrane is.
To determine erythrocyte elasticity, Guedes, et al. looked at AFM tip penetration depth. They found that red blood cells from patients with non-ischemic CHF had a higher average stiffness than cells from the ischemic group and the control group. However, red blood cells from patients with ischemic CHF had higher cell penetration depth. Viscosity studies also showed that cells from non-ischemic patients were higher than ischemic patients and the control group. The cell stiffness may be one of the reasons for a higher incidence of cardiovascular events due to blood clots.
Finally, the AFM studies were correlated with patient clinical data. After twelve months the patients that had a higher binding force between fibrinogen and the erythrocyte receptor were more likely to be hospitalized for a cardiovascular complication than the other two groups. Statistical studies show a correlation between binding strength and hospitalization within the next twelve months.
While this is only a pilot study with a limited number of patients, the authors point out that these results show that AFM is a promising nanotool for looking at the interaction between fibrinogen and erythrocyte binding, which can be used to identify those patients who are at increased risk for a subsequent cardiovascular event.
More information: Ana Filipa Guedes et al. Atomic force microscopy as a tool to evaluate the risk of cardiovascular diseases in patients, Nature Nanotechnology (2016). DOI: 10.1038/nnano.2016.52
Abstract
The availability of biomarkers to evaluate the risk of cardiovascular diseases is limited. High fibrinogen levels have been identified as a relevant cardiovascular risk factor, but the biological mechanisms remain unclear. Increased aggregation of erythrocytes (red blood cells) has been linked to high plasma fibrinogen concentration. Here, we show, using atomic force microscopy, that the interaction between fibrinogen and erythrocytes is modified in chronic heart failure patients. Ischaemic patients showed increased fibrinogen–erythrocyte binding forces compared with non-ischaemic patients. Cell stiffness in both patient groups was also altered. A 12-month follow-up shows that patients with higher fibrinogen–erythrocyte binding forces initially were subsequently hospitalized more frequently. Our results show that atomic force microscopy can be a promising tool to identify patients with increased risk for cardiovascular diseases.
Journal information: Nature Nanotechnology
© 2016 Phys.org