Creating a reduced-fat chocolate that melts in your mouth
Chocolate is divinely delicious, mouthwateringly smooth and unfortunately full of fat. But reducing the fat content of the confection makes it harder and less likely to melt in your mouth. That's why scientists are investigating additives that could reinstate chocolate's delightful properties in these lower-fat treats. Now, researchers report in ACS' Journal of Agricultural and Food Chemistry an analysis that sheds light on how adding limonene could improve lower-fat versions' texture and ability to melt.
Flavor and sweetness make strong contributions to the pleasant experience of eating chocolate, but so do look and feel. Reducing the fat in chocolate, however, often ruins its texture and viscosity. Previous research has shown that adding limonene - a compound found in lemons and oranges - results in a smoother, softer chocolate that melts more easily than typical reduced-fat chocolates. Annelien Rigolle and colleagues at KU Leuven in Belgium sought to investigate exactly how limonene impacts chocolate production. They focused on one part of this process: the crystallization of one of its main ingredients, cocoa butter, which undergoes several important transformations at different times and temperatures.
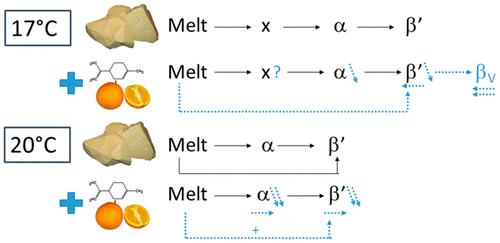
The researchers examined crystallization at 63 °F and 68 °F using differential scanning calorimetry and X-ray diffraction to examine cocoa butter profiles when limonene was added. Surprisingly, they found that adding the compound accelerated cocoa butter crystallization at 63 °F, but inhibited cocoa butter crystallization at 68 °F. Varied concentrations of limonene also affected the crystallization steps of the cocoa butter differently, so they could ultimately affect the texture of chocolate. The study suggests that carefully choosing the amount of limonene and the temperature at which chocolate is processed could lead to a smoother, more luxurious reduced-fat chocolate.
More information: Annelien Rigolle et al. Isothermal Crystallization Behavior of Cocoa Butter at 17 and 20 °C with and without Limonene, Journal of Agricultural and Food Chemistry (2016). DOI: 10.1021/acs.jafc.5b05965
Abstract
Differential scanning calorimetry and real-time X-ray diffraction using synchrotron radiation were used to elucidate isothermal cocoa butter crystallization at 17 and 20 °C in the absence and presence of different limonene concentrations. At 17 °C, a three-step crystallization process was visible for pure cocoa butter, whereby first an unknown structure with long spacings between a 2L and 3L structure was formed that rapidly transformed into the more stable α structure, which in turn was converted into more stable β′ crystals. At 20 °C, an α-mediated β′ crystallization was observed. The addition of limonene resulted in a reduction of the amount of unstable crystals and an acceleration of polymorphic transitions. At 17 °C, the crystallization process was accelerated due to the acceleration of the formation of more stable polymorphic forms, whereas there were insufficient α crystals for an α-mediated β′ nucleation at 20 °C, resulting in a slower crystallization process.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society