April 27, 2016 report
A new Diels-Alder reaction
(Phys.org)—The Diels-Alder reaction is a mainstay in organic chemistry. The reaction traditionally involves a diene and a dienophile. The diene has four carbons that are sp2 hybridized to form pi bonds. The dienophile has two sp2 hybridized carbons. The diene and dienophile undergo a cycloaddition reaction to form a cyclohexene. This reaction can be tweaked to make six-membered rings with a higher oxidation state by removing hydrogens from the reactants to make sp hybridized carbons. For example, rather than an alkene and a diene reacting, the reactants could be an alkyne and an allenyne.
In the current research Teng Wang, Rajasekhar Reddy Naredla, Severin K. Thompson, and Thomas R. Hoye trap an intermediate formed from the removal of five hydrogens from the typical reactants in a [4+2] Diels-Alder cycloaddition. This intermediate, called α,3-dehydrotoluene, is of interest because its oxidation state is analogous to benzyne, and like benzyne, it can be chemically trapped. This allows for functionalization at unique positions on the molecule. Their work appears in Nature.
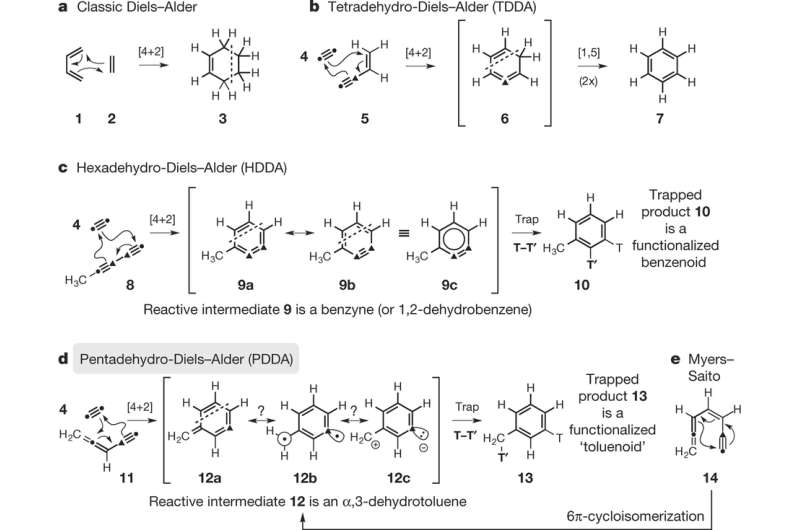
Removal of hydrogens from the diene of a [4+2] Diels-Alder cycloaddition results in either an allenyne, an unsaturated organic molecule that contains both a double and triple bond, or a diyne, a molecules with two triple bonds.
In the case of a tetra-dehydro Diels-Alder reaction, in which four hydrogens are removed, the cycloaddition results in a benzene ring. In the hexa-dehydro Diels Alder reaction (HDDA), a alkyne and a diyne reaction to form a benzyne intermediate, 1, 2-dehydrobenzene. The penta-dehydro Diels Alder reaction (PDDA) involves the reaction of an allenyne an alkyne to produce α,3-dehydrotoluene as an intermediate. The benzyne intermediate from the HDDA reaction and the α,3-dehydrotoluene intermediate from the PDDA reaction are tautomers of each other, and both can be trapped using an appropriate trapping agent. Hoye's group is the first to trap α,3-dehydrotoluene in a PDDA cyclization reaction.
Hoye's group first suspected that this intermediate occurs from a reaction they did with a particular tetrayne at room temperature in a solution of piperidine. α,3-dehydrotoluene has been observed as a result of Myers-Saito cyclization reaction, but prior to this study, has not been isolated as part of a formal cycloaddition reaction. They found that differences between the intermediates in the HDDA and PDDA reaction has to do with a base-promoted rate-limiting step in the PDDA reaction.
Their initial reaction showed that the tetrayne undergoes a piperdine-catalyzed isomerization to produce an allenyne. This then undergoes a PDDA cyclization reaction whose intermediate can be trapped with piperidine, a secondary amine. This mechanism can be generalized to a base-promoted isomerization to produce an allenyne, followed by trapping the intermediate with a protic trapping agent. Hoye's group looked at other primary and secondary amines as well as oxygen-based nucleophiles such as methanol and water, and found that all of them served as good trapping agents for the α,3-dehydrotoluene intermediate.
Hoye's group also found that the PDDA cyclization reaction worked with a nitrile in place of the alkyne on the allenyne reactant. Furthermore, the PDDA cyclization worked with electron withdrawing groups on the nitrile, and the intermediate can be trapped using the same trapping agents as with the allenyne reaction. Notably, nitriles to do not undergo cyclization in HDDA reactions, therefore, its ability to cyclize in PDDA reactions allows for greater diversity in the types of products that can be made from these reactions.
This newly discovered reaction mechanism and isolation of its intermediate allows for the synthesis of several types of organic molecules that would be of interest, including pyridine products. Additionally, it provides insight into the mechanistic differences between the hexa-dehydro Diels Alder reaction and the penta-dehydro Diels Alder reaction, which may be exploited for further studies on cyclization reactions.
More information: Teng Wang et al. The pentadehydro-Diels–Alder reaction, Nature (2016). DOI: 10.1038/nature17429
Abstract
In the classic Diels–Alder [4 + 2] cycloaddition reaction, the overall degree of unsaturation (or oxidation state) of the 4π (diene) and 2π (dienophile) pairs of reactants dictates the oxidation state of the newly formed six-membered carbocycle. For example, in the classic Diels–Alder reaction, butadiene and ethylene combine to produce cyclohexene. More recent developments include variants in which the number of hydrogen atoms in the reactant pair and in the resulting product is reduced by, for example, four in the tetradehydro-Diels–Alder (TDDA) and by six in the hexadehydro-Diels–Alder (HDDA) reactions. Any oxidation state higher than tetradehydro (that is, lacking more than four hydrogens) leads to the production of a reactive intermediate that is more highly oxidized than benzene. This increases the power of the overall process substantially, because trapping of the reactive intermediate can be used to increase the structural complexity of the final product in a controllable and versatile manner. Here we report an unprecedented overall 4π + 2π cycloaddition reaction that generates a different, highly reactive intermediate known as an α,3-dehydrotoluene. This species is in the same oxidation state as a benzyne. Like benzynes, α,3-dehydrotoluenes can be captured by various trapping agents to produce structurally diverse products that are complementary to those arising from the HDDA process. We call this new cycloisomerization process a pentadehydro-Diels–Alder (PDDA) reaction—a nomenclature chosen for chemical taxonomic reasons rather than mechanistic ones. In addition to alkynes, nitriles (RC≡N), although non-participants in aza-HDDA reactions, readily function as the 2π component in PDDA cyclizations to produce, via trapping of the α,3-(5-aza)dehydrotoluene intermediates, pyridine-containing products.
Journal information: Nature
© 2016 Phys.org