Evolution of hermaphroditic nematode proves deterministic maternal effects can give offspring head start in life
Thank your mothers: A research collaboration between UC Santa Barbara and L'École Normale Supérieure in Paris has proven that deterministic maternal effects can give offspring a better start on life.
"It's the first observation of experimental evolution of a maternal effect in response to a changing environment," said UCSB evolutionary biologist Stephen Proulx, an author of the article that appears in PLOS Biology. The results of the study, which involved the evolution of a tiny hermaphroditic worm, shed light on strategies employed by organisms in response to changes in their habitats.
Unlike genotypic effects, which dictate traits in offspring based on inherited genes, parental (in this case, maternal) effects influence offspring phenotypes—physical traits—during embryonic development. From the point of view of the developing embryo, Proulx said, these traits depend on the quality of their environment as they grow and develop. In mammals, for instance, the mother's body size and nutritional status will affect her growing fetus' size. Her ability to transfer nutrients or secrete one hormone or another during pregnancy also affects the offspring's development.
"In mammals, the placenta is an organ for maternal effects," said Proulx. As the organ through which nutrients, hormones and waste pass between mother and fetus, the placenta and its health and functioning have a direct impact on the baby's development.
In a wider sense, mom's own genetics—which also dictate her body size, health and other things that could affect her developing infant—contribute to maternal effects.
"There are whole papers written on 'What do we mean when we say maternal effect,' " Proulx said of the ongoing effort to fully define the phenomenon.
As part of natural selection, these influences, which may be randomized or deterministic, can be evolutionary strategies that benefit individuals and affect species survival. Variations in traits within even the same generation and same family may be direct responses to the environment experienced by the mother—or the egg—and could, with optimal timing, ensure that offspring are specifically better adapted to the environment they could experience (deterministic).
Other maternal effects could ensure that at least a few of several siblings will live to adulthood, given traits rather more arbitrarily provided to them (randomized) that could increase their chances of survival—also known as "bet hedging."
"The idea of bet hedging is that if the environment is unpredictable then you might produce a variety of offspring that could do well in various environments because you can adjust the concentration of different resources," Proulx said. Taken from gambling, bet hedging essentially attempts to decrease the chance of a complete loss of offspring, however some of the population is expected to do poorly or not survive.
For all the theory, however, a direct demonstration of the benefit of maternal effects has been missing, according to the researchers.
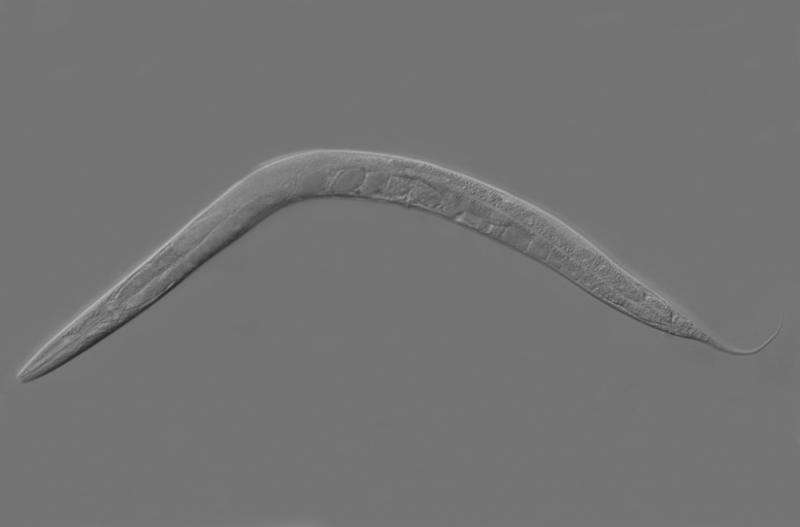
Enter Caenorhabditis elegans, a tiny hermaphroditic nematode with a short developmental cycle, the ability to easily produce many offspring and a fully mapped genome. To study the type and impact of maternal effects under varying environmental conditions, Parisian collaborators Snighadip Dey and Henrique Teotónio subjected generations of the worm to different patterns of normal oxygen conditions (normoxia) and low-oxygen environments (anoxia), while the nematodes were still in the egg stage, and then monitored the traits produced in the offspring for 60 generations.
They found that when the environment changed in a predictable manner—in this case, alternated every generation between anoxic and normoxic embryonic development—mothers that had experienced normoxic conditions early on tended to provision their young (which were oxygen-deprived as embryos) with more glycogen, in effect equipping the embryos with tools to survive the stress of oxygen deprivation.
"Maybe it just means that a gene got turned on when mom was developing herself that then causes more glycogen to get put into each egg," Proulx said of the adaptation. "What's really cool is that this is a seven-day life cycle so mom experienced normoxia as an embryo and then five days later, when she goes to lay her own eggs, she adjusts how much glycogen gets put in it. This is a memory that lasts basically her whole life span, that affects how she provisions her offspring."
In contrast, added Proulx, it didn't work the other way: The non-oxygen-deprived offspring of mothers who experienced anoxia as eggs did not fare any worse despite the mother's early "memory" of no oxygen.
Additionally, over the course of the experimental evolution, there also developed a trade-off between fertility and hatchability in predictable-environment offspring that had survived anoxia: While fewer eggs were laid by them as adults, the likelihood of survival of the eggs that were laid—hatchability—increased.
Meanwhile, in the unpredictable scenarios in which the anoxic and normoxic conditions fluctuated between successive generations, bet-hedging would have been the expected strategy to deploy for survival.
"Under the random unpredictable conditions, we didn't see an increase in offspring survivorship in either normoxic or anoxic environments," Proulx said. "We didn't see the evolution of bet-hedging, which could have happened, and it's something we originally expected might happen." Lack of this development across successive generations suggests that randomized maternal effects are not only less common, they are also less environment-responsive and perhaps only on rare occasions do they lead to adaptations to fluctuating environments. On the other hand, the study suggests also that once deterministic maternal effects have evolved, they could be adaptive to a variety of fluctuating environments.
Looking ahead, Proulx and team are taking a wider view of the phenomenon.
"We want to understand how the history of evolving maternal effects will affect populations as they experience future changes in the environment," he said. "If they've evolved these adaptive maternal effects, are they going to do better if now the environment starts to change more, and also whether information can be transferred over more than one generation."
More information: Snigdhadip Dey et al. Adaptation to Temporally Fluctuating Environments by the Evolution of Maternal Effects, PLOS Biology (2016). DOI: 10.1371/journal.pbio.1002388
Journal information: PLoS Biology
Provided by University of California - Santa Barbara