Uncovering secrets of elastin's flexibility during assembly
Elastin is a crucial building block in our bodies - its flexibility allows skin to stretch and twist, blood vessels to expand and relax with every heartbeat, and lungs to swell and contract with each breath. But exactly how this protein-based tissue achieves this flexibility remained an unsolved question - until now.
This material has a remarkable combination of flexibility and durability: elastin is one of the body's most long-lasting component proteins, with an average survival time comparable to a human lifespan.
During a person's life, the elastin in a blood vessel, for example, will have gone through an estimated two billion cycles of pulsation.
A team of researchers at the University of Sydney, MIT in the United States and at the University of Manchester in the United Kingdom has carried out an analysis that reveals the details of a hierarchical structure of scissor-shaped molecules that gives elastin its remarkable properties.
The findings are published today in the journal Science Advances, in a paper by the University of Sydney postdoctoral research associate Dr Giselle Yeo and Professor Anthony Weiss in the Faculty of Science and Charles Perkins Centre, with co-authors including MIT graduate student Anna Tarakanova and Professor of Civil and Environmental Engineering Markus Buehler.
Elastin tissues are made up of molecules of a protein called tropoelastin, which are strung together in a chain-like structure and which Professor Weiss and his team have been studying in the lab for many years. In this work, they collaborated with Professor Buehler and Ms Tarakanova at MIT, who have specialised in determining the molecular structure of biological materials through highly detailed atomic-scale modeling. Combining the computational and laboratory approaches provided insights that neither method could have yielded alone, team members say.

While the study of elastin has been going on for a long time, Professor Weiss says this particular paper was exciting on a number of levels: because of synchrotron imaging done by team member Clair Baldock at the University of Manchester, the research revealed the shape and structure of the basic tropoelastin molecules. But these were snapshots - still images that could not illuminate the complex dynamics of the material as it forms large structures that can stretch and rebound.
Those dynamic effects were revealed through the combination of computer modeling and laboratory work. "It's really by combining forces with these three groups" that the details were pieced together, Professor Weiss said.
Ms Tarakanova explained that in Professor Buehler's lab, "we use modeling to study materials at different length scales, and for elastin, that is very useful, because we can study details at the sub-molecular scale and build up to the scale of a single molecule." By examining the relationship of structure across these different scales, she said, "we could predict the dynamics of the molecule".
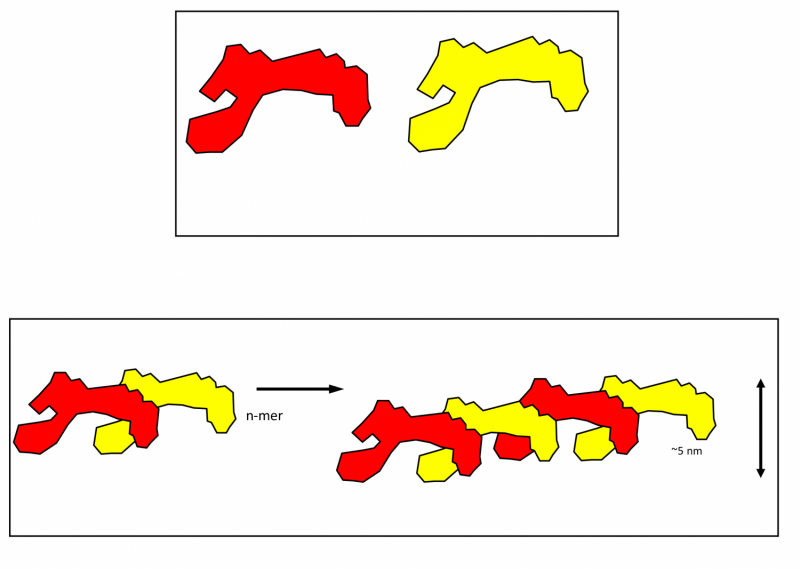
The dynamics turned out to be complex and surprising, Professor Weiss said. "It's almost like a dance the molecule does, with a scissors twist - like a ballerina doing a dance." Then, the scissors-like appendages of one molecule naturally lock onto the narrow end of another molecule, like one ballerina riding piggyback on top of the next. This process continues, building up long, chain-like structures.
These long chains weave together to produce the flexible tissues that our lives depend on - including skin, lungs, and blood vessels. These structures "assemble very rapidly," Professor Weiss said, and this new research "helps us understand this assembly process".
A key part of the puzzle was the movements of the molecule itself, which the team found were controlled by the structure of key local regions and the overall shape of the protein.
The team tested the way this flexibility comes about by genetically modifying the protein and comparing the characteristics of the modified and natural versions. They revived a short segment of the elastin gene that has become dormant in humans, which changes part of the protein's configuration. They found that even though the changes were minor and just affected one part of the structure, the results were dramatic. The modified version had a stiff region that altered the molecule's movements.
This helped to confirm that certain specific parts of the molecule, including one with a helical structure, were essential to contributing to the material's natural flexibility.That finding in itself could prove useful medically, the team says, as it could explain why blood vessels become weakened in people with certain disease conditions, perhaps as a result of a mutation in that gene.
While the findings specifically relate to one particular protein and the tissues it forms, the team said the research may help in understanding a variety of other flexible biological tissues and how they work.
"The integration of experiment and modelling in identifying how the molecular structure endows materials with exceptional durability, elasticity, and studying how these materials fail under extreme conditions, yields important insights for the design of new materials that replace those in our body, or for materials that we can use in engineering applications in which durable materials are critical," Professor Buehler said.
"We are excited about the new opportunities that arise from this collaboration and the potential for future work, because designing materials that last for many decades without breaking down is a major engineering challenge that nature has beautifully accomplished, and on which we hope to build."
More information: Subtle balance of tropoelastin molecular shape and flexibility regulates dynamics and hierarchical assembly, Science Advances, , dx.doi.org/10.1126/sciadv.1501145
Journal information: Science Advances
Provided by University of Sydney