Jawless fish brains more similar to ours than previously thought
Researchers at the RIKEN Evolutionary Morphology laboratory and other institutions in Japan have shown that complex divisions in the vertebrate brain first appeared before the evolution of jaws, more than 500 million years ago. Published in Nature, the study shows that two elements of brain genoarchitecture thought to be unique to jawed vertebrates are actually present in two jawless fish—the hagfish and lamprey.
Most living vertebrate species have jaws, a development thought to have occurred sometime in the Paleozoic era. Jawed vertebrates—including humans—share many developmental characteristics that have remained unchanged for millennia. The brain's basic developmental plan was thought by many scientists to have reached completion in jawed vertebrates because the brains of lampreys and hagfish—the only jawless fish that remain alive today—seem to lack two key domains.
Recent evidence brought this into question, and as the only lab in the world able to study hagfish embryos, the RIKEN team led by Shigeru Kuratani was in a unique position to use techniques derived from developmental biology to tackle this critical issue.
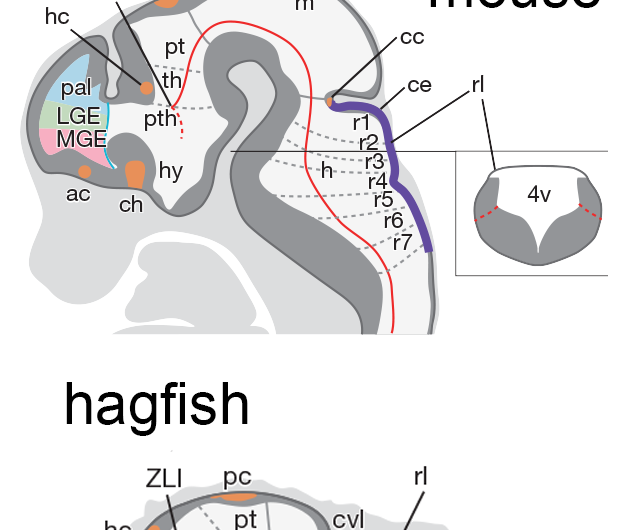
The vertebrate brain develops from a neural tube that is divided into sections. The development of each section is very specific, and is controlled by the expression of particular genes at very precise times and locations. These gene-expression patterns—or the genoarchitecture—are highly conserved in jawed vertebrates. Lampreys—a type of jawless fish—appear to lack two brain regions common to jawed vertebrates—the cerebellum and a region called the medial ganglionic eminence, or MGE, from which the pallidum and cortical interneurons originate.
In jawed vertebrates, the MGE develops from a forward section of the neural tube that expresses Nkx2.1 and Hedgehog genes, and the cerebellum develops from a region called the rhombic lip that expresses Pax6. In hagfish, the team found a region in the correct location that expresses both Nkx2.1 and a Hedgehog gene that was identified for the first time in this study. This indicated that the hagfish brain does indeed have an MGE region. Similarly, although hagfish do not have a true cerebellum, the team was able to identify a clear rhombic lip region that expresses Pax6.
At this point, the team was confident that the brains of both hagfish and jawed vertebrates contain similar developmental patterning.
"The problem was that lampreys had not yet been shown to have a similar patterning," explains Kuratani. "The shared pattern of brain development between hagfish and jawed vertebrates raised the possibility that the apparently primitive brain of the lamprey is simply a lamprey-unique characteristic."
Additionally, without an answer to the lamprey question, these shared patterns could be interpreted as changes that independently occurred in each lineage—jawed and jawless—after they split from each other.
To address the issue, the team reinvestigated the lamprey, and discovered several new Nkx2.1 genes expressed in the correct location, but did not find any Hedgehog expression, indicating that the lamprey MGE is slightly different from that found in jawed vertebrates. Then the team looked at lamprey larvae and found a rhombic-lip like region that expresses Pax6B, albeit slightly differently than in hagfish or jawed vertebrates.
"We found that jawed-vertebrate patterning was more similar to the hagfish than to lampreys," says Kuratani, "and the evidence indicates that this is likely due to secondary evolutionary changes in lamprey evolution, rather than changes unique to jawed vertebrates."
"With these new findings from hagfish and lampreys, we have shown that both of the extant jawless-fish species have a rhombic lip and an MGE —the sources of the cerebellum, pallidum, and GABAergic interneurons in jawed vertebrates. This firmly places the development of these genoarchitectural patterns back to a common ancestor shared by jawless and jawed vertebrates."
More information: Fumiaki Sugahara et al. Evidence from cyclostomes for complex regionalization of the ancestral vertebrate brain, Nature (2016). DOI: 10.1038/nature16518
Journal information: Nature
Provided by RIKEN













.jpg)