Researcher unlocks genetic secrets to birds' behavior, evolution
Nearly 30 years of fieldwork coupled with cutting-edge technology has yielded important genetics insight recently published by the academic journal Current Biology and reviewed in Science Magazine.
For Elaina Tuttle, it's like winning an Oscar.
"I think of all the researchers throughout the years, all the field assistants and graduate assistants who have been collaborators. It's nice because even though they may not be in authorship, a little part of them is in there," said Tuttle, professor of biology and associate dean of graduate programs at Indiana State University.
"I think about my first years, not knowing what we're doing, and now I think about the well-oiled machine we are with the white-throated sparrow project."
Tuttle and fellow State biology Professor Rusty Gonser have led 27 years' research into white-throated sparrows at Cranberry Lake in New York. Their efforts have led to greater understanding of the birds' supergene, its apparent degradation and the species' evolution.
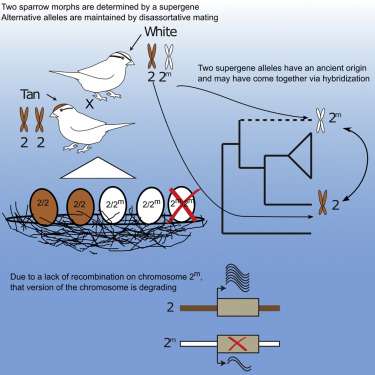
The white-throated sparrow has two morphs—tan and white—that differ in color and behavior because of a chromosomal inversion.
"There's this large chunk of chromosome two that is rotated. It acts like a supergene and prevents any sort of recombination. It links those genes so there are no changes in them, generation after generation after generation," Tuttle said.
With these supergenes, the good traits are passed on together to offspring and never unlinked.
"If you're really aggressive and it's good to be aggressive with your bright color, that's always linked together and passed on together," she said. "It prevents the breakup of good combinations. But it can also harbor bad combinations."
By sequencing the genome, Tuttle and the team of researchers—including Sycamore graduate students Marisa Korody and Adam Betuel—have identified more than 1,000 genes and where they are located.
"What we find is that in the white morph, (the supergene is) degrading," she said. "Not only is the white supergene degrading, but it's dragging the tan supergene along with it. It's called purifying selection—it's getting rid of all those bad combinations. You're going to be left with only the good combinations."
The findings have important parallels to human sex genes.
"So, kind of like the X and the Y sex chromosomes in humans, the X chromosome is really large, and the Y chromosome is really tiny. They think it was (once equal)," she said.
They also unlocked key components to the birds' evolution—the white morph is newer, and the tan morph is the ancestral form.
"By using these evolutionary analysis tools, we were able to figure out there was probably hybridization with some extant or extinct species a long time ago," she said.
The morphs breed with the opposite—a tan male with a white female and so on—which perpetuates the species and its health.
"We were able to show that when white pairs with white, there's detriment," Tuttle said. "They have genetic problems. They also have increased promiscuity - they're not so happy together. It does pay to pair with the opposite morph."
Typically, researchers have genomics data or they have field data. "You don't often have both integrated into a nice story together," she said.
And that field data dates back nearly three decades.
"We can go back in history and do the genomics from samples from 1988 and see how genes change over time. It's a really rich database," she said.
Until genome sequencing capabilities caught up with Tuttle, she says it was like having a black box that needed to be unlocked.
"I knew there were secrets in there, and we're just beginning to see," she said. "Now, the data can be mined by us and other researchers because it's on a public database."
As questions are answered, even more are asked.
"I'm excited. There's so many other things we can find out—how genes arise, how they're maintained, how they work," she said. "We're even thinking about doing epigenetics, which is how genes are turned on or off. One of the future projects we're looking at is how stress turns on certain genes. That's a big thing in human disease, as well. Stress turns on certain genes that cause disease later in life. It's a good model for human behavior as well."
Countless Indiana State students and postdocs, as well as students from other universities—domestic and international—have participated in Tuttle's research. And this latest paper will become the basis of limitless future work.
"It made it even more rewarding having to go through the struggle of waiting for the technology to get there," she said. "This is probably my pinnacle paper. Finding the behavioral polymorphism was the beginning of my career with the sparrows. I'm going to still have papers, but this is a big turning point."
More information: Elaina M. Tuttle et al. Divergence and Functional Degradation of a Sex Chromosome-like Supergene, Current Biology (2016). DOI: 10.1016/j.cub.2015.11.069
Journal information: Science , Current Biology
Provided by Indiana State University