Can CRISPR help edit out female mosquitos?

The recent discovery of the first male-determining factor in mosquitoes, combined with the gene-editing capabilities of the CRISPR-Cas9 system, could be used to bias mosquito populations from deadly, blood-sucking females toward harmless, nectar-feeding males, thus helping to prevent the spread of mosquito-borne diseases. Two experts outline the advantages and challenges of such approaches in a Review published Feb. 17, 2016 in Trends in Parasitology, part of a special issue on vectors.
"We are at a turning point both in our understanding of how mosquitoes determine whether to become a male (a good choice for us) or a female (trouble for us), as well as our ability to permanently modify wild populations using gene drive techniques," says Zach Adelman, an entomologist at Virginia Tech. According to Adelman and co-author Zhijian Tu, also of Virginia Tech, genetic engineering holds promise for preventing the spread of mosquito-borne diseases such as dengue, malaria, yellow fever, and Zika.
Genetic strategies to control dengue fever based on the release of sterile, transgenic mosquitoes are currently underway and have been successful where attempted. However, these techniques are expensive and impractical because they require long-term releases and are difficult to implement at a large scale. A potentially more effective and less costly approach might be to drive maleness genes with the CRISPR-Cas9 system, which has shown promise as an easy, efficient, and precise approach to introduce mutations at virtually any genomic site of interest in a wide range of organisms, including mosquitoes.
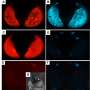
This proposed strategy recently came one step closer to reality. In a high-profile study published in Science last May (DOI: 10.1126/science.aaa2850), Adelman and Tu reported their discovery of the first male-determining factor in mosquitoes. Expression of this so-called M factor, a sex-determination gene called Nix, in female embryos triggered the development of external and internal male genitalia. Nix was both required and sufficient to initiate male development in Aedes aegypti—a major carrier for dengue, yellow fever, Zika, and chikungunya viruses.
"This discovery sets the stage for future efforts to leverage the CRISPR-Cas9 system to drive maleness genes such as Nix into mosquito populations, thereby converting females into males or simply killing females," Tu says. "Either outcome would help to reduce mosquito populations and improve sex separation procedures, which are required in any genetic strategy to prevent the accidental release of disease-transmitting females into wild populations."
But many questions need to be answered before such gene drive systems can be applied in the field. For one, it is still not clear how Nix controls sex determination in mosquitoes, and whether M factors are conserved across species that transmit different diseases. Moreover, the effectiveness and long-term stability of CRISPR-based gene drive systems in mosquitoes remain unknown.
There are also several ethical concerns. For example, the accidental release of just a few mosquitoes during testing may be sufficient to establish gene drive systems in the wild, and it is not clear how to remove an introduced gene from a study area if needed. Meanwhile, fears of bioterrorism have prompted some to call for the prohibition of public discussion of the details of gene drive approaches. Because clear regulatory and ethical guidelines have not yet been established, the National Academies of Sciences, Engineering, and Medicine is currently developing recommendations for the responsible conduct of gene drive research in non-human organisms.
Given these concerns, the approach will most likely be first applied at locations where the disease burden is high, other control measures are not successful, public sentiment is supportive, and the regulatory structure in place is navigable, Adelman says. "Moving forward, partnerships with supportive governments, local collaborators and a willing public will be crucial to establishing field-based testing in areas that are most impacted by mosquito-borne diseases."
More information: Trends in Parasitology, Adelman and Tu: "Control of mosquito-borne infectious diseases: Sex and gene drive" dx.doi.org/10.1016/j.pt.2015.12.003
Journal information: Trends in Parasitology , Science
Provided by Cell Press