January 12, 2016 feature
Researchers attempt to uncover the origins of water's unusual properties
(Phys.org)—In many ways, water behaves very differently than other liquids do, and with important consequences: It's widely thought that water's unusual properties were essential for the development of life on Earth. One prime example is the fact that ice floats. Unlike other substances, frozen water is less dense than liquid water, and this anomaly allows fish and other aquatic lifeforms to survive in the water under a frozen layer of protective ice during cold periods. Water also has a high heat capacity, meaning it can absorb and release a large amount of heat while undergoing very little change in temperature. This property helps many living organisms maintain a relatively stable body temperature, and it also provides a pleasant climate in Europe due to the warm Gulf Stream current.
It's widely known that water's anomalous properties are related to its hydrogen bonds, which cause liquid water to arrange itself in a highly ordered way because of the attraction between the hydrogen atoms in one water molecule and the oxygen atoms in adjacent molecules. However, researchers do not completely understand how water's unique hydrogen-bonded structure leads to its anomalous properties.
In a review paper published in Nature Communications, physicists Anders Nilsson and Lars G.M. Pettersson from Stockholm University have pulled together the results from dozens of papers published over the past several years that have investigated water's molecular structure, often with the help of cutting-edge experimental tools and simulations.
In their interpretation of the data, the researchers have proposed a picture of water in which its unique properties arise from its heterogeneous structure.
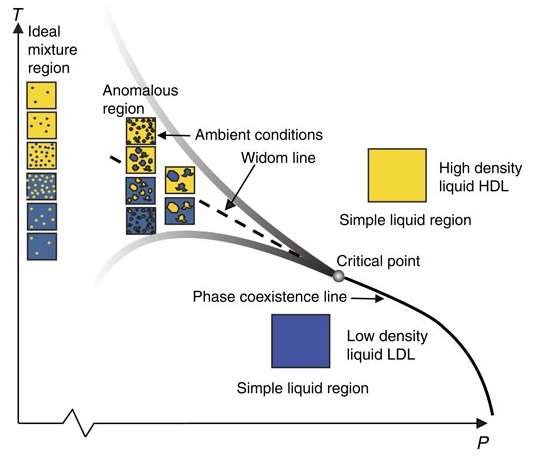
The researchers propose that, in the pressure (P) and temperature (T) region where water exhibits its anomalous behaviors (the funnel-like region in the P-T phase diagram above), water coexists in two different types of structures: a highly ordered, low-density structure with strong hydrogen bonds, and a somewhat smashed, high-density structure with distorted hydrogen bonds. The origins of water's anomalous properties arise because these two types of structures are constantly fluctuating between one another in this heterogeneous phase, resulting in many small spatially separated regions of different structures.
"It is the fluctuations between high-density liquid and low-density liquid that give rise to the anomalous properties," Nilsson told Phys.org.
Take water's large heat capacity, for instance. As Nilsson explained, heat capacity is related to entropy fluctuations, and entropy is in turn related to the number of possible ways the available energy can be distributed in the system. Fluctuations between the low- and high-density local structures increase the magnitude of the entropy fluctuations and, consequently, the heat capacity.
Outside of the funnel-like region, water stops behaving anomalously and begins behaving more like other liquids. In these regions, water's structure is homogeneous, existing as only the low-density structure below the funnel-like region and only the high-density structure above the funnel-like region.
While the new paper combines many years of data into one cohesive picture, many questions still remain. One unanswered question, for instance, is why does water's anomalous region occur at the same temperatures and pressures that sustain life? It seems likely that water's anomalous region served to place constraints on the conditions required for life to exist. A better understanding of this overlap could have implications for understanding life on a fundamental level.
Many other questions also require further investigation. The researchers explain that their proposed interpretation is a very simple picture, and it does not tell the exact degree of heterogeneity in water in the anomalous region, nor does it describe the nature of the boundaries between the spatially separated fluctuating regions.
Another area of investigation that has challenged researchers is the so-called "no-man's land" region, which refers to the possibility of liquid water at temperatures below 232 K (-42 °C and -42 °F) but above 150 K (-123 °C, -189 °F). It's extremely challenging to cool liquid water to these cold temperatures quickly enough so that it can be probed, even for a moment, before it turns to solid ice. In 2014, scientists successfully cooled liquid water to 5 K below no-man's land and measured its structure using an X-ray laser—the Linac Coherent Light Source at the SLAC National Accelerator Laboratory. Experiments like these are needed to answer the question of whether water can exist in a liquid state in this region, which would help complete the P-T phase diagram.
In the future, the researchers plan to investigate how water's structural fluctuations contribute to other intriguing phenomena.
"We will focus on using X-ray lasers to probe the supercooled regime and search for the liquid-liquid transition and second critical point," Nilsson said. "We are also developing probe dynamics using the femtosecond time resolution of the X-ray lasers, allowing eventually to probe the dynamics of these structural fluctuations. There might be some intricate coupling of time and space on various length and time scales that are still unknown.
"We hope that others will also bring other techniques involving time resolved two-dimensional IR spectroscopy, NMR and neutron scattering to further investigate water along these lines. Also further refinement of theoretical approaches to both calculate experimental observables but also to simulate water."
More information: Anders Nilsson and Lars G.M. Pettersson. "The structural origin of anomalous properties of liquid water." Nature Communications. DOI: 10.1038/ncomms9998
Journal information: Nature Communications
© 2016 Phys.org





















