Step tsunamis wash away crystal impurity effects

Under laboratory conditions, mineral growth is a well understood physical process, and has been for a number of decades. In nature however, outside of the controlled confines of a lab, mineral formation is far more complex and scientists are struggling to understand how crystalline minerals can form at all. Workers in the fields of mineralogy, geochemistry and biomineralization have long faced an apparent contradiction: Natural minerals are formed under conditions that should, according to existing models of crystal growth, prohibit their formation.
The apparent contradiction arises because, first, natural minerals tend to form under low supersaturation conditions. This is a direct consequence of the fact that in a natural setting, supersaturation is only gradually increased over geological timescales, yielding crystals that grow close to equilibrium. Second, these natural environments are typically heavily 'polluted' with respect to the main crystallizing species. These pollutants, or impurities, are potent inhibitors of crystal growth since they block sites where new atoms or molecules prefer to attach onto the crystal surface. Such low-supersaturation/low-purity conditions should block crystal growth . At least, that is the prediction of established crystal growth theories.
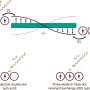
Researchers from the Vrije Universiteit Brussel and the Université Libre de Bruxelles now contest that picture in their recent work on crystal growth featured in the 1-8 January Issue of Physical Review Letters. Using kinetic Monte Carlo simulations, they show that a particular class of steps (unfinished layers on the crystal surface), so-called macrosteps, can provide an escape route from impurity-induced surface poisoning. Macrosteps are the result of dozens or even hundreds of layers growing at once, and comprise abundant surface features on a wide range of crystals. Lutsko, et al show that macrosteps can bypass the paralyzing impurity effect by a combination of two effects.
First, macrosteps boast strength in numbers. The grouping of a large number of steps induces a crowding effect that pushes the system outside of kinetic arrest. Effectively, macrosteps can reduce the induction time required to push past regions rich in impurities, which is the rate-limiting step of growth in this regime, by multiple orders of magnitude. Macrosteps can, in that sense, be regarded as tsunamis that wash away the impurities bound to the surface. The second way by which macrosteps can undo the impurity effect has to do with their structure as a function of height. Sleutel and coworkers show that as the macrostep height increases, they transform from densely packed step trains into facets with simple crystallographic indices. These facets facilitate the nucleation of three-dimensional clusters on the intersection area between the macrostep facet and the vicinal crystal face. Such 3D clusters locally breach the impurity-dense regions that cause pinning by incorporating and overgrowing them.
The tsunami comparison is again apt: Macrosteps are huge step waves that can simply overflow the areas that are poisoned by impurities. After dissecting the physical principles that underlie this new mechanism of growth recovery for a simplified scenario, Lutsko et al demonstrate the generality of their results by incorporating a wide range of crystal growth effects. They compare the simulation results with a published experimental study on the KDP/Fe system (Land, 1999, Nature; Thomas, 2004, PRL): the first system known to exhibit macrostep mediated growth recovery, but for which a physical model was still missing. The Monte Carlo simulations reproduce all the key experimental observations as well as expose the missing physics needed to rationalize the observed kinetic dependencies.
More information: James F. Lutsko et al. Step Crowding Effects Dampen the Stochasticity of Crystal Growth Kinetics, Physical Review Letters (2016). DOI: 10.1103/PhysRevLett.116.015501
Journal information: Physical Review Letters , Nature
Provided by Université libre de Bruxelles