Crystal structures of human TIM members: Ebola virus entry-enhancing receptors
Since first identified in 1976, Ebola virus has caused a total of 24 outbreaks involving 1,716 cases according to the World Health Organization (WHO) statistics. The current outbreak in West Africa, with the first case identified in March 2014, is the largest and most complex Ebola outbreak. It includes more than 28,000 infection cases and over 11,000 deaths as of December 2015. The WHO's emergency committee unanimously agreed that the current outbreak—centered in Guinea, Liberia, and Sierra Leone—was "an extraordinary event and a public health risk to other states", and declared the outbreak a "public health emergency of international concern".
Ebola virus is a group of enveloped viruses that can infect both primates and humans. They are taxonomically categorized into five species, including Zaire (ZEBOV), Sudan (SUDV), Tai Forest (TAFV), Bundibugyo (BDBV) and Reston (RESTV) ebolavirus. The extremely high mortality rate related to Ebola virus infection (25 percent to 90 percent in past outbreaks) makes the virus one of the most deadly pathogens. Over the past few decades, scientists paid great efforts to have a deeper understanding of this most virulent pathogen and revealed a series of receptors that play important roles in cell entry of Ebola virus.
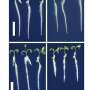
Human T-cell immunoglobulin and mucin domain (hTIM) receptor was recently identified as the entry-enhancing receptor for various enveloped viruses, including Ebola virus. This hTIM-mediated entry is highly dependent on the immunoglobulin variable (IgV)-like domain of the receptor, which specifically recognizes envelope phosphotidylserine (PS). Of the three hTIMs (hTIM-1, -3, and -4), however, neither their IgV domain structures nor the PS-binding basis is available thus far, though their murine homologues have been structurally well defined. More than that, functional details of how the hTIMs work in the processing of cell entry of Ebola virus are not well known.
In an article titled "Crystal structures of human TIM members: Ebolavirus entry-enhancing receptors," researchers, led by Prof. George Fu Gao, showed that lentivirions pseudotyped with either the 1976-ZEBOV or the 2014(C05)-ZEBOV glycoproteins (GPs) exhibit similarly enhanced cell entry via hTIM-1, though the two GPs contain multiple residue variance.
As expected, this hTIM-mediated enhancement of Ebola virus infection is highly dependent on the PS-binding capacity of the receptor. Furthermore, hTIM-4 with a mucin domain comparable to that of hTIM-1 in length, but not hTIM-3 that contains only limited numbers of mucin repeats, efficiently enhance pseudovirus infection, demonstrating, as previously reported, an important role of the hTIM mucin domain in facilitating the virus entry. They further demonstrated that hTIMs do not directly interact with ZEBOV GP, regardless of whether their mucin domains are present or not. They also solved the IgV domain structures of all the three hTIM family members and revealed individual regional features that were not observed in any of the mouse homologues. These atomic structural observations, together with the solved complex structure of phosphoserine bound to hTIM-4, shed perceptive light on the atomic interactions between PS and hTIMs.
Taken together, the structural and functional data provided by Prof. George Fu Gao's group paved the way for a better understanding of the hTIM-mediated Ebola virus entry.
More information: Han WANG et al. Crystal structures of human TIM members: Ebolavirus entry-enhancing receptors, Chinese Science Bulletin (Chinese Version) (2015). DOI: 10.1360/N972015-01255
Provided by Science China Press