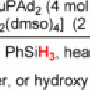November 13, 2015 report
Palladium-catalyzed C-H activation of primary amino alcohols
(Phys.org)—Many synthetic schemes to produce active biomolecules or pharmaceuticals begin with simple starting materials that can serve as a generalizable backbone to several chemical reactions. Ideally, these starting materials can be functionalized in many ways to produce a large variety of compounds.
One class of molecules that has proved difficult to functionalize is primary aliphatic amines. There are several biomolecules and pharmaceuticals that require a primary amine as part of its structure. However, amines are highly reactive while aliphatic carbons are not. The metal catalyst employed to activate a C-H bond would typically react with the primary amine instead. One example is cyclometallation, a palladium-catalyzed reaction that activates C-H bonds in the presence of alcohols and secondary amines.
Jonas Calleja, Daniel Pla, Timothy W. Gorman, Victoriano Domingo, Benjamin Haffemayer, and Matthew J. Gaunt from the University of Cambridge have found a generalizable solution. They demonstrate that the C-H bond of an aliphatic amino alcohol can be functionalized with a variety of substituents via a palladium-catalyzed cyclometallation reaction by temporarily functionalizing the amino alcohol such that the amine is sterically hindered from reacting with palladium. They used their synthetic pathway to make over sixty different compounds, including a known pharmaceutical for multiple sclerosis, fingolimod. Their work appears in Nature Chemistry.
Typically chemists use some kind of protecting group to keep a reaction from occurring at a different reactive site from the target site. However, protecting groups can be difficult to remove without damaging the molecule, costing time and resulting in lower yields. Gaunt, et al.'s research began with an amino alcohol containing a primary –OH, a primary –NH2 and a fully substituted carbon atom. This is a common piece to more complex structures seen many biologically important molecules. They wanted to functionalize the aliphatic region while still keeping a primary amine and alcohol.
The problem is in how readily the amine coordinates to metals. In palladium-catalyzed C-H bond activation, the amine coordinates with the palladium, and when two amines coordinate with the metal, it forms a stable bis(amine) Pd(II) complex, precluding any reaction on the target carbon.
To keep the primary amines from forming the bis(amine) Pd(II) complex, Gaunt, et al. reacted the amino alcohol with a "steric tether", in this case, cyclohexanone. This tether temporarily bridges the oxygen from the primary alcohol to the primary amine, making it a secondary amine. Prior research by this group showed that secondary amines do not coordinate to palladium as readily, allowing for the reaction to occur.
They tested their mechanism with an acetoxylation reaction. They found that hydrogen bonding between the secondary amine hydrogen and the acetate ligand helped prevent the formation of the bis(amine) Pd(II) complex. Additionally, they determined that the chemical oxidant, iodosobenzene diacetate, was needed to optimize the reaction and obtain good yields. The results were that their reaction scheme is broadly applicable on a variety of amino alcohols with a diverse set of substrates on the substituted carbon.
The next step was to see if this reaction mechanism worked by functionalizing the C-H bond with other biologically relevant substituents. They tried a C-H arylation of a variety of amino alcohols. One difference between this scheme and the acetoxylation is that using cis-3,5-dimethyl cyclohexanone as the steric tether produced better yields, likely because the methyl-substituted cyclohexanone added additional steric hindrance to the system. Importantly, this reaction worked with aryl groups that had a variety of substituents, including electron withdrawing and electron donating groups.
Gaunt, et al. then tested their synthetic mechanism by conducting a palladium-catalyzed C-H carbonylation. By subjecting several amino alcohol derivatives to a carbon monoxide atmosphere, they were able to form pyrrolidinones. Pyrrolidiones are five-membered heterocyclic ketones, comprised of a secondary amine and are found in many biologically relevant and pharmaceutical compounds. They found that this reaction functions best in the presence of silver salt and an air mixture of 6.25% CO.
Finally, Gaunt, et al. conducted a palladium-catalyzed C-H alkenylation of several amino alcohol derivatives. Alkenylation is traditionally a very difficult carbon-carbon bond-forming reaction, but by using a similar strategy as the carbonylation reaction, they were able to produce several pyrrolidines in good yields. They found that this reaction required silver acetate, trifluoroethyl acrylate and Li3PO4 to obtain good yields.
This research demonstrates how a simple aliphatic amino alcohol can be converted into a broad range of complex amines and has implications for optimizing and streamlining many synthetic schemes that require C-H activation in the presence of a primary amine. The implications of this research are demonstrated in their model synthesis of the pharmaceutical reagent, fingolimod (Commercial name: Gilenya), in which they successfully made the compound in high yield. Their route is more streamlined than typical ways to make fingolimod.
More information: Jonas Calleja et al. A steric tethering approach enables palladium-catalysed C–H activation of primary amino alcohols, Nature Chemistry (2015). DOI: 10.1038/nchem.2367
Abstract
Aliphatic primary amines are a class of chemical feedstock essential to the synthesis of higher-order nitrogen-containing molecules, commonly found in biologically active compounds and pharmaceutical agents. New methods for the construction of complex amines remain a continuous challenge to synthetic chemists. Here, we outline a general palladium-catalysed strategy for the functionalization of aliphatic C–H bonds within amino alcohols, an important class of small molecule. Central to this strategy is the temporary conversion of catalytically incompatible primary amino alcohols into hindered secondary amines that are capable of undergoing a sterically promoted palladium-catalysed C–H activation. Furthermore, a hydrogen bond between amine and catalyst intensifies interactions around the palladium and orients the aliphatic amine substituents in an ideal geometry for C–H activation. This catalytic method directly transforms simple, easily accessible amines into highly substituted, functionally concentrated and structurally diverse products, and can streamline the synthesis of biologically important amine-containing molecules.
Journal information: Nature Chemistry
© 2015 Phys.org