Ionic and covalent drug delivery
Researchers at Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences compared three different drug delivery models based on ionic liquids. Scientists have developed a powerful API-IL concept to realize structural diversity and dual-action pharmaceuticals. The study, published in ACS Medicinal Chemistry Letters, introduces new drug delivery concepts using ionic and covalent molecular level forces.
The human body is composed of numerous types of molecules, both simple and complex, and all fundamental processes in a living body occur in water solutions. Therefore, for a drug to work, it must dissolve in body liquids, which are primarily water. Polymorphism of solid substances, a well-known problem of drug delivery, is the ability of solid drugs to form several different crystal structures (polymorphs). Polymorphs may differ in properties like biological activity, and in addition, their formation is difficult to control.
The problem of crystal polymorphs can be solved by liquid substances or solutions, as their medical usage is more predictable and reliable. Thus, many researchers investigate solubilization of existing active pharmaceutical ingredients. It was previously established that transformation of a poorly soluble substance into a salt may substantially increase solubility.
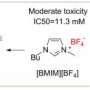
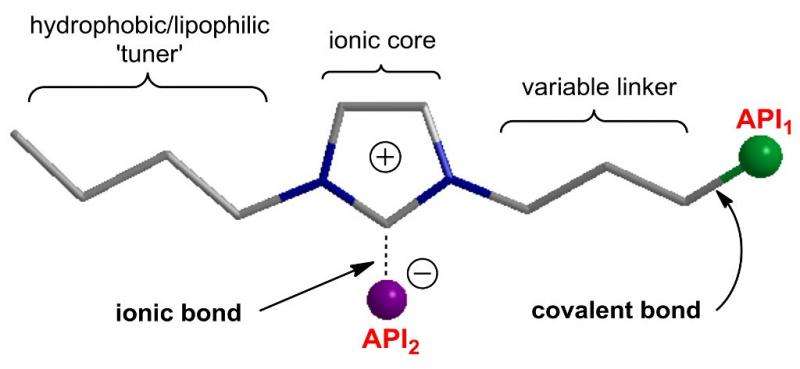
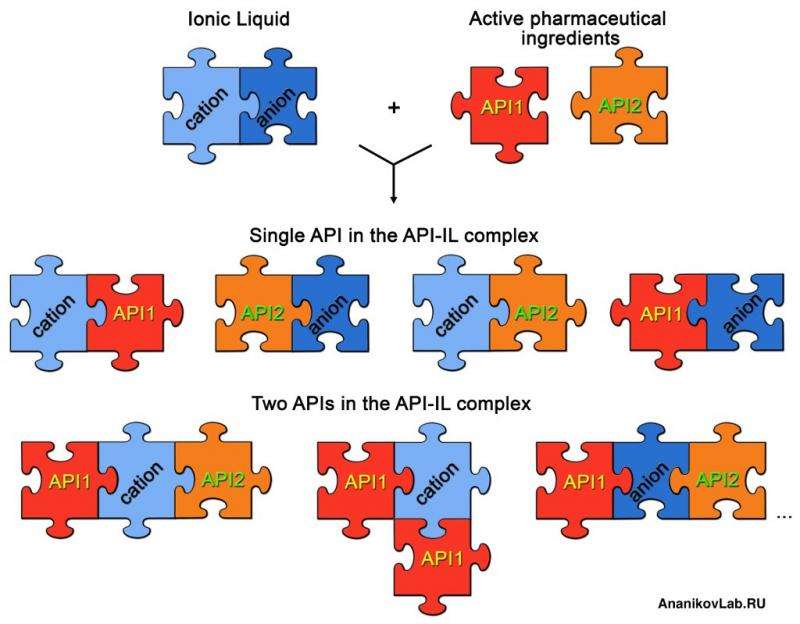
The scientists led by Professor Ananikov proved that ionic liquids are excellent candidates to connect organic molecules to salt structures. Currently, there are three techniques to introduce active pharmaceutical ingredients into the ionic liquids: (i) as an anion or a cation (ionic bond); (ii) via a covalent linkage (covalent bond); and (iii) using both ionic and covalent binding.
The proposed drug development platform has the following valuable advantages:
- Tunable hydrophobicity/lipophilicity for adjusting the ability to penetrate through cell membranes and other biological barriers.
- Ionic core, which implies the ability to modulate the strength of ionic binding. Ionic liquids are highly tunable and can be readily optimized to connect with a variety of pharmaceutical ingredients.
- Variable linker for regulating the distance between the ionic core and active pharmaceutical ingredient. This linker may contain a specific site of enzymatic cleavage for target release of active pharmaceutical ingredient in the place of action.
The researchers used salicylic acid (a well-known anti-inflammatory compound) as a model drug and incorporated the molecule of salicylic acid into ionic liquids. The scientists studied solubility and biological activity of salicylic acid-containing ionic liquids toward human fibroblasts and colorectal adenocarcinoma cells. Indeed, salicylic acid retained its activity in ionic liquid form. At the same time, it demonstrated dramatically higher water solubility in the complex with ionic liquid, as compared to the pure salicylic acid.
The study points out the important advances of API-IL concept in drug development and pharmaceuticals. Variability and ability to access a diversity of molecular combinations should be taken into account. A highly promising application is the production of 'dual-action' drugs bearing two different active pharmaceutical ingredients with ionic and covalent binding. This approach may allow complex treatment targeting different potential causes of the pathology.
More information: "Cytotoxic Activity of Salicylic Acid-Containing Drug Models with Ionic and Covalent Binding." ACS Med. Chem. Lett., Article ASAP September 28, 2015 DOI: 10.1021/acsmedchemlett.5b00258
Journal information: ACS Medicinal Chemistry Letters
Provided by Zelinsky Institute of Organic Chemistry