New gene drive technology evokes hopes and fears
The idea of introducing a novel gene into a few individuals that then spreads through an entire population sounds like a premise for science fiction. And yet fiction can be prophetic.
Cornell researchers have used mathematical models to illuminate the promises – and potential problems – of a new genome editing mechanism, called a gene drive.
The mechanism has been long discussed but only recently demonstrated in fruit flies in labs at the University of California, San Diego, using genome-editing technology called CRISPR. The gene drive mechanism may allow scientists to control malarial mosquitoes or pesticide-resistant pests, for example, by using CRISPR to introduce a mutation (allele) into a few individuals in a population and have that mutation quickly spread through the entire population. But researchers fear such gene drives could have unintended consequences, such as spreading into an unintended species.
Cornell researchers report the need to thoroughly understand the population dynamics and behavior of artificial gene drive systems, according to a recent paper in the journal Genetics. To do this, they created a simple mathematical model to test how quickly and how far an introduced allele might spread.
"The time for these CRISPR alleles to spread and become fixed in a population is on the order of tens of generations," said Rob Unckless, the paper's first author and a postdoctoral research fellow in the Department of Molecular Biology and Genetics. "That's incredibly fast." Under natural selection, a beneficial gene may take hundreds of generations to reach high frequencies in a population, he added.
CRISPR technology was first derived from a bacterial immune response that uses RNA to protect itself against plasmids and viruses by breaking specific DNA sequences in the pathogen's genome. With the CRISPR technology, researchers have co-opted that system to edit any gene, allowing them to make precise genetic changes for a desired effect.
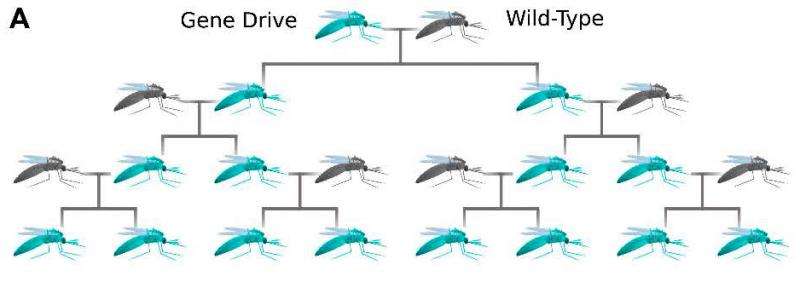
Each individual within higher species has two copies of every gene, one from each parent. UCSD researchers figured out how to use an autonomous CRISPR system to edit a gene and change the corresponding allele so that it carries the new genetic edit. Once an embryo has a CRISPR allele, that individual organism will develop two exact copies, which it passes on to its offspring. The mechanism then continues through subsequent generations, thereby spreading an allele rapidly through a population.
Now researchers can take a gene that has a fitness cost, such as male sterility, and move it through a population, something very unlikely under natural selection.
The mathematical model showed that an introduced allele is more likely to become fixed in a population when that allele efficiently converts its corresponding allele into a match, and when the fitness costs to the individual are weak.
Under natural conditions, a single copy of any mutation is almost always lost. But Cornell researchers found a single mutation introduced in one individual spread to the entire population close to one-third of the time, assuming that the CRISPR allele perfectly converted its corresponding allele, and the introduced allele carried a fitness cost where an individual produced 40 percent fewer offspring.
"That's one of the things that is scary, if you imagine that one of these alleles gets into a population that you don't want it in," Unckless said. For example, a CRISPR gene that controls mosquitos that carry dengue may jump to a related species that perhaps doesn't feed on humans. A bigger concern would be if a CRISPR gene somehow transferred through a mite or wasp to honey bees, Unckless said.
"There is so much we don't know, but it's also so promising," Unckless said. "Nobody has done the modeling, so we are working on this."
More information: Modeling the Manipulation of Natural Populations by the Mutagenic Chain Reaction, Genetics July 30, 2015 genetics.115.177592
Journal information: Genetics
Provided by Cornell University