Strategies to decrease bacterial colonization
Among the bacterial infections that are most difficult to treat, chronic infections associated with bacterial biofilms are one of the most hazardous. Bacterial biofilms are densely packed communities of microbial cells surrounded with secreted polymers. In her doctoral thesis, chemist Shoghik Hakbyan has studied ways to decrease the bacterial colonization at Umeå University in Sweden.
A common example is the slimy layer that can form inside the drains from your sink. These biofilms can be formed on all surfaces and cause serious problems for our society especially in health care. It has been reported that biofilms are responsible for approximately 80 per cent of all microbial infections and cause 100,000 deaths annually. Thus, control of bacterial growth and colonization is of critical importance.
Antibiotics are the usual treatment in case of bacterial infections. However, traditional antibiotics are becoming increasingly inefficient due to the development of antibiotic resistance in bacteria. Furthermore, by forming biofilms, bacteria survive under harsh conditions and are up to 1,000 times less susceptible to most antibiotics. Thus, the easiest way to combat biofilms is to prevent bacterial adhesion and biofilm formation on surfaces.
The prevention of biofilm formation can be done either by loading surfaces with antibacterial compounds, or by fabricating surfaces that inhibit bacterial adhesion. Several strategies are being developed to inhibit bacteria adhesion, and one of the most promising is the attachment of polymer brushes to form antibacterial surfaces. A polymer brush is a layer of polymer chains tethered with one end to a surface and stretched away from the substrate resembling a forest of bottle brushes with the side chains sticking out as bristles on a bottle brush.
In Shoghik Hakobyan's thesis, a class of small organic molecules called hydrazones was used as antibacterial compounds. These substances are able to disarm bacteria without killing them. Since bacteria stay alive, the evolutionary pressure for developing resistance can be considered lower than for bactericidal compounds. Furthermore, the hydrazones bind metal ions, such as gallium ions.
"Gallium ions kill both planktonic and biofilm bacteria, but it has to be in a complex with other molecules to work most efficiently and not form insoluble solid aggregates," says Shoghik Hakobyan.
One part of her doctoral thesis is about the chemical and biological properties of gallium-hydrazone complexes. Two of the most bioactive ones were studied and the results showed that they form very strong complexes with gallium. This indicated that these complexes would be stable at physiological conditions and would deliver gallium ions to bacterial cells. Moreover, these complexes have interesting biological effects and could suppress pathogenic actions of bacteria or inhibit biofilm formation.
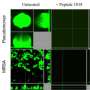
The second part of the thesis related to surface modifications with different types of polymer brushes and testing of their antibacterial properties. For long-lasting antibiofilm surfaces, it is very important to generate surfaces that repel for example proteins, in order to reduce the deposition of cell debris on the surface and subsequent loss the bioactivity.
"My results show that the brushes containing equal amounts of positively and negatively charged units strongly suppressed biofilm formation, as well as reduced attachment of proteins and bacteria. Furthermore, I have developed and optimized a strategy to anchor the bioactive hydrazone molecules to these brushes in order to make biologically active surfaces," says Shoghik Hakobyan.
More information: "Antivirulent and antibiofilm salicylidene acylhydrazide complexes in solution and at interfaces," umu.diva-portal.org/smash/get/ … 49571/FULLTEXT02.pdf
Provided by Umea University