Hydrogen for all seasons

Ludwig Maximilian University of Munich chemists have developed novel porous materials called "covalent organic frameworks", which provide a basis for the design of polymeric photocatalysts with tunable physical, chemical and electronic properties.
Chemical systems that are capable of generating hydrogen gas by light-activated scission of water molecules (often termed artificial photosynthesis) represent a promising technology for the efficient storage of solar energy. However, the systems that have been developed so far suffer from various drawbacks, and intensive efforts are underway to discover alternative procedures that are both more practical and efficacious. Chemists led by Professor Bettina Lotsch, who has dual appointments in the Department of Chemistry at LMU and the Max Planck Institute for Solid State Research in Stuttgart now introduce a new class of porous organic materials that can be used as the basis for molecularly tunable photocatalysts for light-driven production of hydrogen gas. The researchers report their findings in the new issue of the online journal Nature Communications.
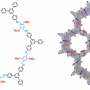
Lotsch and her colleagues are interested in the properties and practical applications of so-called covalent organic frameworks. These materials are composed of layers of regular two-dimensional molecular networks synthesized from simple organic precursors, and they exhibit a number of features that facilitate photocatalytic processes. "They form crystalline and porous semiconductors, whose chemical properties can be precisely tuned for a given application," as Bettina Lotsch explains. They are already under investigation as possible matrices for the storage of gases and for applications in sensor technology, and also have considerable potential in the field of optoelectronics.
More efficient and more economical
In collaboration with the group led by Christian Ochsenfeld, Professor of Theoretical Chemistry at LMU, Lotsch and her team have been exploring the potential of such porous polymers as photocatalysts. In their latest work, they chose so-called triphenylarenes as the basic subunits of their model matrix. "The great advantage of this class of materials is that the chemical and physical properties of the network can be readily engineered for different applications, simply by altering the structure of the precursors," says Vijay Vyas, a postdoc in Bettina Lotsch's group at the Max Planck Institute for Solid State Research. "This flexibility allowed us progressively to modulate their ability to produce hydrogen. Their performance parameters in this context are comparable to those of established photocatalysts based on carbon nitride and oxides." The planar layers of the new set of compounds are synthesized from hydrazine and a series of aromatic trialdehydes. In the resulting structure, the trialdehyde subunits are linked together by azine (=N-N=) bridges to form two-dimensional lattices.
Metal-based photocatalysts are often expensive to make and difficult to modify. "But since the properties of COFs can be readily and specifically altered, their performance characteristics can also be manipulated at will," says Frederik Haase, a member of Bettina Lotsch's group. They therefore provide a combination of features which make them ideal as a basis for the development of environmentally friendly and economical photocatalysts.
Bettina Lotsch summarizes the results of the study as follows: "We have now demonstrated, at the molecular level, that the structural, morphological and optoelectronic properties of covalent organic frameworks can be precisely tuned so as to maximize their photocatalytic activity." The advances made by the LMU chemists thus promise to make solar energy even more attractive as a future source of sustainable energy.
More information: A tunable azine covalent organic framework platform for visible light-induced hydrogen generation, Nature Communications 6, Article number: 8508 DOI: 10.1038/ncomms9508
Journal information: Nature Communications
Provided by Ludwig Maximilian University of Munich