Superionic conductor for fuel cells
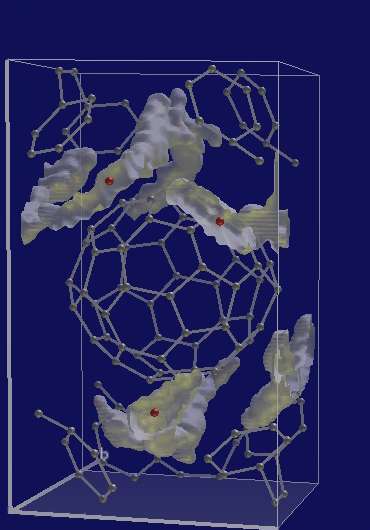
An alternative material for use in fuel cells has been probed by the ILL, with the aim of understanding its peculiar bonding structure and complex behaviour.
Buckminsterfullerene, or C60 buckyballs, were first generated in the mid-80s, but despite a high research profile and the Nobel Prize in Chemistry that followed, few direct applications of the cage-like molecule have been found. However, when atoms of alkali-metals such as lithium (Li) are added to the cage, the buckyballs undergo polymerisation, forming long chains that create a material with a range of new properties. Despite being a solid, Li4C60 displays an ionic conductivity comparable to that of liquid electrolytes, even at room temperature. And this has led to suggestions that this material could find use in future fuel-cells or batteries.
In this project, neutrons and magnetic resonance techniques were used to pinpoint the location of the carbon atoms involved in the intermolecular bonds, and to reveal the Li positions in the lattice. This process involved a series of neutron diffraction and spectroscopy experiments, carried out on the ILL Super D2B two-axis diffractometer, and the IN4C and IN1BeF spectrometers. In parallel with this, the team used theoretical lattice calculations to reveal the polymer structure, and to characterise its dynamic behaviour.
The agreement between structural models and the experimental data confirms the coexistence of unusual bonding among the buckyballs and suggests that charge transfer between lithium and the C60 cage is responsible for this behaviour. The information concerning the positioning of the lithium between the cages provides important insight into the origin of the high mobility of these ions. This mobility, in addition, seems tributary to the cage motion. In fact hybrid modes, i.e. modes for which lithium ions and adjacent buckyballs move in communion, could be identified at relatively low frequencies. This observation makes the team believe that the ion mobility may be highly dynamic with the motion of the buckyballs "pushing" the lithium ions along and thus playing an important role in the material's temperature-dependent ionic conductivity.
ILL Scientist Stephane Rols said "This work will help in understanding the role of the guest lattice structure and plasticity in the material's high Li-ionic conductivity. Development of a full description of the phenomenon is a necessary first step to the design of solid material with controlled ionic conductivity. This would generate new generations of Li-batteries that integrate non-liquid ionic exchange membrane, potentially making them safer."
More information: "Structure and dynamics of the fullerene polymer Li4C60 studied with neutron scattering", Physical Review B, 92, 014305 (2015). DOI: 10.1103/PhysRevB.92.014305
Journal information: Physical Review B
Provided by Institut Laue-Langevin




















