New simple proteins play active role in cellular function
Yale scientists have developed simple new proteins almost devoid of chemical diversity that still play a surprisingly active and specific role in cellular function, causing cells to act like cancer cells, they report Aug. 10 in the Proceedings of the National Academy of Sciences.
The wide array of protein function, upon which all life depends, is due to the specific sequences of large groups of amino acids that typically number in the hundreds or thousands. The side chains of these amino acids display considerable chemical diversity, giving rise to an enormous number of protein structures such as enzymes that carry out biochemical reactions and carrier proteins such as hemoglobin, which transport oxygen to our tissues.
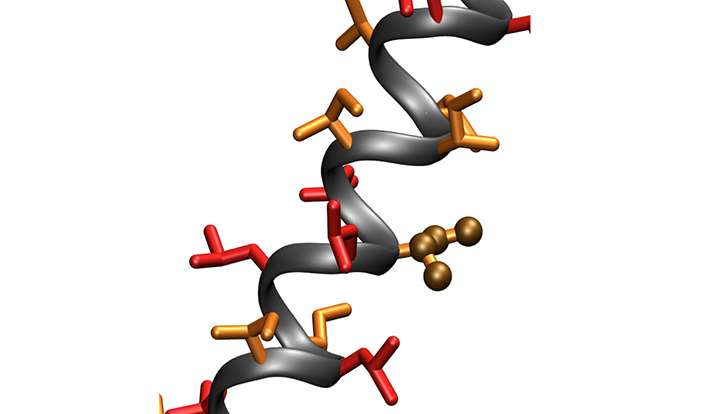
Prompted by studies of a short viral protein that spans cell membranes and causes tumors, the Yale team devised a series of synthetic membrane proteins just 26 amino acids long. Remarkably, they report, they have even built these proteins from sequences of just two amino acids with very similar side chains. Despite this extraordinary simplicity, a tiny fraction of these sequences display biologic activity and can be isolated by genetic selection, said the study's senior author Dr. Daniel DiMaio, the Waldemar Von Zedtwitz Professor of Genetics and deputy director of Yale Cancer Center.
"We have built the simplest proteins possible, and not only are they active, they are specific. They can find a single target in cells and activate it, resulting in uncontrolled cell growth," DiMaio said. "We wonder if there are similar proteins in cells that have been overlooked because of their simplicity, some of which might cause cancer. It might be time to rethink what is considered an active protein."
DiMaio added that synthetic biology efforts, including the development of active agents and potentially new drugs, could be advanced by insights gleaned from studying these simple proteins.
More information: "Biologically active LIL proteins built with minimal chemical diversity." PNAS 2015 ; published ahead of print August 10, 2015, DOI: 10.1073/pnas.1514230112
Journal information: Proceedings of the National Academy of Sciences
Provided by Yale University