Self-assembled aromatic molecular stacks, towards modular molecular electronic components
Being able to effectively tune the electron-transport properties of a single-molecule has been a long-standing issue towards the crystallization of molecular electronics, where individual molecules mimic the behavior of common electronic components as a true alternative to conventional silicon devices. To functionalize electron transport properties, each and every individual molecule must be precisely aligned in place with sub-nanometer precision. In that sense, stacks of self-assembled aromatic components in which non-covalently bound π-stacks act as replaceable modular components are promising building blocks.
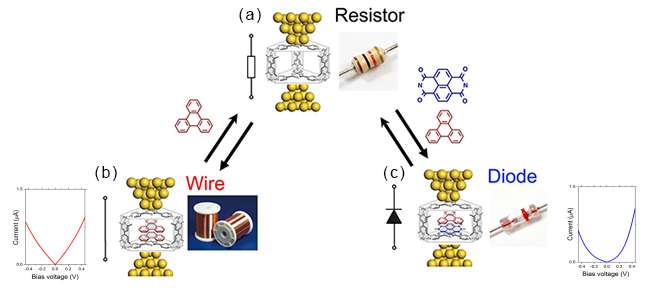
Here we describe the electron-transport properties of aromatic stacks aligned in a self-assembled cage, using a scanning tunneling microscope (STM) based break-junction method. Both identical and different modular aromatic pairs are non-covalently bound and stacked within the molecular scaffold leading to a variety of fascinating electronic functions. The empty cage presents a low electronic conductance (10–5 G0) characteristic of resistors (Figure a) while the insertion of identical molecular pairs results in a marked conductance increase (10–3–10–2 G0, G0 = 2e2/h) mimicking the behavior of electronic wires (Figure b). On the contrary, when different molecular pairs are inserted into the scaffold, electronic rectification (rectification ratio 2–10) characteristic of a diode can be observed (Figure c).
Theoretical calculations demonstrate that this rectification behavior originates from the different stacking order of the internal aromatic components with respect to the direction of the electron-transport, and the corresponding lowest unoccupied molecular orbital conduction channels localized on one side of the molecular junctions.
This study paves the way for the development of molecular electronic devices with tunable electronic functions.
More information: "Rectifying electron-transport properties through stacks of aromatic molecules inserted into a self-assembled cage." Journal of the American Chemical Society 137, 5939–5947 (2015) DOI: 10.1021/jacs.5b00086
Journal information: Journal of the American Chemical Society
Provided by Tokyo Institute of Technology