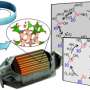
Nanocatalysts improve processes for the petrochemical industry
With the goal of advancing the oil, mining and energy industries, as well as counteracting the emission of greenhouse gases, nanotechnologist Hector Barron Escobar designs more efficient and profitable nanomaterials.
The Mexican, who lives in Australia, studies the physical and chemical properties of platinum and palladium, metals with excellent catalytic properties that improve processes in petrochemistry, solar cells and fuel cells, which, because of their scarcity, have a high and unprofitable price—hence, the need to analyze their properties and make them long-lasting.
Structured materials that the researcher designs can be implemented in the petrochemical and automotive industries. In the first, they accelerate reactions in the production of hydrocarbons, and in the second, nanomaterials are placed in catalytic converters of vehicles to transform the pollutants emitted by combustion into less harmful waste.
PhD Barron Escobar, who majored in physics at the National University of Mexico (UNAM), uses virtual supercomputers to interact with atomic models under different conditions before applying findings to the real world.
Barron recounts how he came to Australia with an invitation of his doctoral advisor, Amanda Partner with whom he analyzed the electronic properties of gold in the United States.
Using computer models in the Virtual Nanoscience Laboratory (VNLab) in Australia, he creates nanoparticles that interact in different environmental variables such as temperature and pressure. He also analyzes their mechanical and electronic properties, which provide specific information about behavior to produce the best working conditions. Together, these data serve to establish appropriate patterns or trends in a particular application.
The work of the research team serves as a guide for experts from the University of New South Wales in Australia, with which they cooperate, to build nanoparticles with specific functions. "This way we perform virtual experiments, saving time, money and offer the type of material conditions and ideal size for a specific catalytic reaction, research which would otherwise cost a lot of money," Barron Escobar comments.
Currently, he designs nanomaterials for the mining company Orica, because in this industry, explosives need to be controlled in order to avoid damaging the minerals or the environment.

Research also applies to the creation of fuel cells. The catalysts designed by Barron make it possible to produce more electricity without polluting.
Additionally, the researchers enhance the effectiveness of catalytic converters in petrochemistry, where these materials help accelerate oxidation processes of hydrogen and carbon, which are present in all chemical reactions when fuel and gasoline are created. "We can identify the ideal particles for improving this type of reactions."
The nanotechnology specialist also seeks to analyze the catalytic properties of bimetallic materials like titanium, ruthenium and gold, as their reaction according to size, shape and its components.
Provided by Investigación y Desarrollo