New fluorescent polymer makes deformation visible

A new type of polymer can show that it has changed shape. After exposure to UV light, the chain-like molecules emit a different colour of light. This opens a new pathway for research into how viruses function in a cell and how minor damage in rubbers and plastics can accumulate and lead to rupture. The new polymers were developed by researchers at Wageningen University, who published an article on their findings in the Journal of the American Chemical Society on 12 August 2015.
A polymer can be compared to a necklace of small molecules that are chemically linked together. Polymers are the basis of a huge variety of natural and artificial materials, from skin, hair and DNA, to the simplest and most advanced plastics. The properties of these polymer materials are largely determined by their spatial structure, also known as 'conformation'.
Polymers can be as straight as uncooked spaghetti, but can also occur as a tangle of cooked spaghetti. Polymer chains resist changes to their conformation, for example when they are stretched. This spring-like effect provides elasticity to rubbers, flexibility to plastics and strength to the cytoskeleton of the cell. Therefore, to change the conformation of a polymer, force must be applied to the molecule. But figuring out the exact conformation of a polymer is particularly difficult, especially if the polymers are surrounded by many other substances, such as in a cell.
Fingerprint
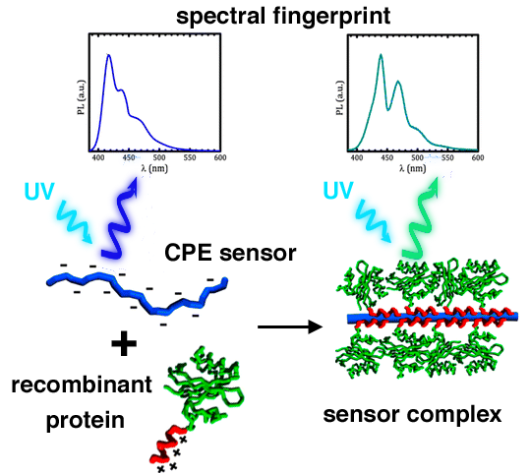
A team of researchers from the Physical Chemistry and Soft Matter Group of Wageningen University, led by Joris Sprakel, has designed a new kind of polymer that 'reports' its spatial configuration to the researchers through the light it emits. PhD candidate Hande Cingil carried out the work on the water-soluble semiconducting polymers, which the researchers have called conjugated polyelectrolytes (CPEs).
Luminescent polymers have existed for some time. They change colour as their conformation changes. A special feature of the CPE polymers is that nuances can be observed in these colour changes. Following irradiation with UV light, the existing polymers emit a colour spectrum that looks like the profile of a mountain with a flat top. But the new polymers have their own 'fingerprint': they show specific peaks in the spectrum. In addition, these peaks shift as the spatial structure changes, for example, if the material in which they are incorporated is stretched. As a result, the novel polymers can detect very small forces on the nanoscale.
Artificial virus
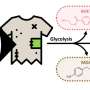
In their publication in the prestigious Journal of the American Chemical Society, the Wageningen chemists demonstrate the functioning of their CPE polymers. For this purpose they used a protein that was designed by their colleagues in Wageningen, Renko de Vries and Martien Cohen Stuart. The protein is a highly simplified version of an artificial virus; like a biological virus, it binds to DNA and subsequently encapsulates it. Sprakel: "In our experiment, the CPE was encapsulated by the simplified artificial virus protein, giving it a rigid layer, which caused the polymer to change shape. Using simple and non-invasive light spectroscopy, this encapsulation process can now be studied in detail."
Rupture
The novel polymers can be used for many purposes. Groups of molecules can be attached to the polymers for specific applications, such as the detection of proteins or toxins. Thanks to the discovery it is possible to study changes in conformation, also deep inside complex substances and materials, in an entirely new way. For example, it offers an improved method for determining exactly how viral proteins stretch and fold to encapsulate DNA, or how very minor damage to polymeric materials gradually accumulates and eventually causes the materials to rupture.
The researchers are currently working on fundamental research that goes beyond showing whether a polymer chain has stretched: they aim to show exactly where in the chain this stretching occurred.
More information: "Monitoring protein capsid assembly with a conjugated polymer strain sensor." Journal of the American Chemical Society, 12 August 2015, DOI: 10.1021/jacs.5b05914
Journal information: Journal of the American Chemical Society
Provided by Wageningen University