June 30, 2015 report
Substrates change nanoparticle reactivity
(Phys.org)—Nanoscale materials tend to behave differently than their bulk counterparts. While there are many theories as to why this happens, technological advances in scanning tunneling microscopy (STM) have allowed researchers to investigate many of these causes by looking at the properties of a single nanoparticle. One area that requires further investigation is how substrate topology affects nanoparticle reactivity. Using the well-known interaction between carbon monoxide (CO) and substrate-supported palladium (Pd) nanoparticles, researchers from the London Centre for Nanotechnology and University College London have demonstrated, for the first time, that the topology of the titanium dioxide (TiO2) substrate affects Pd nanoparticle reactivity. Their findings are reported in the Proceedings of the National Academy of Sciences.
Chemical reactions on nanoparticles are dependent on properties that are not normally encountered on regular materials. One important property is the physical orientation of the nanoparticles with each other. In bulk reactions, bond distance and particle size are fixed features and the environment plays a more pivotal role. This is not the case in nanoscale materials where greater or smaller distances, orientation, and morphology between two particles can affect nanoparticle reactivity.
One way to control nanoparticle orientation is to deposit metal nanoparticles on a substrate surface. It is only recently that scientists have begun to understand the role that substrate morphology plays in nanoparticle properties, including reactivity. In the case of Pd on a TiO2 substrate, Yim, et al. found that the strain within Pd nanoparticles changes when they are formed across the TiO2 substrate steps. This strain affects Pd reactivity.
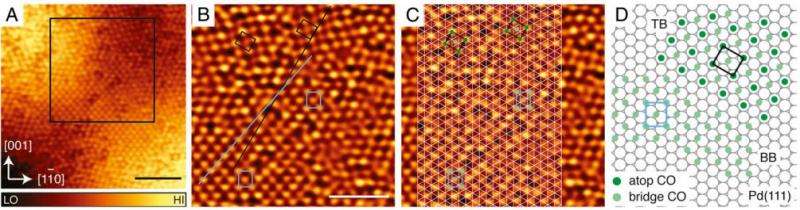
CO adsorbs onto the Pd nanoparticles with (111) top facet in three known adsorption sites at concentrations greater than 0.5 monolayers. As the concentration of CO increases, CO will fill bridge sites on palladium nanoparticles as well as hollow sites. It will then fill a combination of atop (directly on top of palladium nanoparticles) and hollow sites. However, where the Pd nanoparticle lattice curves, CO will bond in different sites.
STM analysis shows that the Pd curvature is due to growth across the step on the TiO2 substrate. This leads to the Pd nanoparticles layering over the substrate steps in way reminiscent of laying a piece of carpet over a step. This leads to curved Pd nanoparticles described as having top (111) facets.
To understand how the curved Pd morphology changes its reactivity, STM studies were performed after 0.5 monolayers of CO were adsorbed onto the Pd surface. Analyses showed that there were different regions of CO. One region has CO molecules occupying bridge sites as is expected when CO is adsorbed on to a Pd/TiO2 system. Another region, however, showed an atop-bridge configuration, which had not been reported for Pd(111) surfaces. Investigation of another step island showed that CO molecules occupy a combination of atop- and face- centered cubic hollow sites, another unobserved configuration for this system.
Computational analyses showed that this difference in CO registry on the curved Pd nanoparticles is probably due to particle strain across the steps. Particle strain has been shown in other systems to affect adsorption of molecules on nanoparticles, although these systems do not show particle strain as localized as on the curved Pd nanoparticles across the TiO2 step islands shown in the work by Yim, et al. The Pd layer seems to elongate across the step edge and shrink in the direction perpendicular to the step edge, causing tensile strain and compression strain. This strain changes how CO interacts with Pd. CO typically binds to Pd at a 90-degree angle and has a double bond character. However, in these new orientations CO is 21.4-degrees from the normal of the Pd nanoparticle and displays a single bond character.
This research has two important implications for nanoparticle-substrate interactions: First, it shows how substrate morphology plays a role in nanoparticle reactivity. Namely, "carpet growth" over step islands probably happens in other systems and may explain changes in reactivity. Second, changing the substrate morphology may be a way to tune nanoparticle reactivity.
More information: "Influence of support morphology on the bonding of molecules to nanoparticles" PNAS, DOI: 10.1073/pnas.1506939112
Abstract
Supported metal nanoparticles form the basis of heterogeneous catalysts. Above a certain nanoparticle size, it is generally assumed that adsorbates bond in an identical fashion as on a semiinfinite crystal. This assumption has allowed the database on metal single crystals accumulated over the past 40 years to be used to model heterogeneous catalysts. Using a surface science approach to CO adsorption on supported Pd nanoparticles, we show that this assumption may be flawed. Near-edge X-ray absorption fine structure measurements, isolated to one nanoparticle, show that CO bonds upright on the nanoparticle top facets as expected from single-crystal data. However, the CO lateral registry differs from the single crystal. Our calculations indicate that this is caused by the strain on the nanoparticle, induced by carpet growth across the substrate step edges. This strain also weakens the CO–metal bond, which will reduce the energy barrier for catalytic reactions, including CO oxidation.
Journal information: Proceedings of the National Academy of Sciences
© 2015 Phys.org