New type of drug can target all disease-causing proteins
Current drugs block the actions of only about a quarter of known disease-causing proteins, but Yale University researchers have developed a technology capable of not just inhibiting, but destroying every protein it targets.
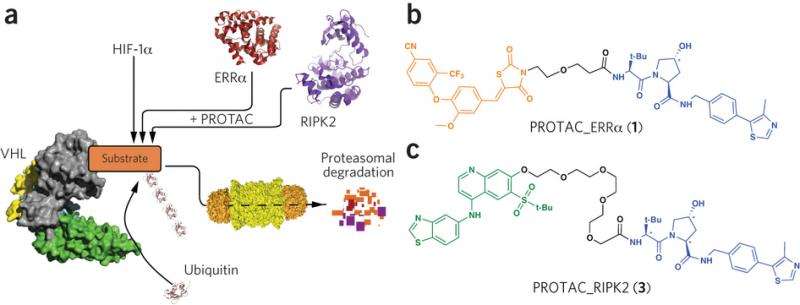
The new type of drug, called Proteolysis Targeting Chimeras (PROTACs), also can continue to destroy mutant proteins in mouse tumors, according to a new study published June 10 in the journal Nature Chemical Biology.
"This new drug modality culminates a decade of work in the field by my lab," said Craig Crews, the Lewis B. Cullman Professor of Molecular, Cellular, and Developmental Biology and senior author of the paper, which was done in collaboration with scientists from GlaxoSmithKline and Arvinas, LLC.
Almost all current drugs are small molecules designed to fit into the folds of disease-causing proteins and inhibit their function. High doses are often needed to ensure that protein function is blocked sufficiently to produce therapeutic results, which in turn can produce harmful side effects.
In contrast, PROTACs engage the cells' own protein degradation machinery to destroy targeted proteins by tagging them for removal and can do so multiple times, meaning it can work at lower doses, the authors say. This suggests this new type of drug has not only the potential to target proteins that are not currently "pharmaceutically vulnerable" but could do so safely, Crews said.
"This is a game-changer for drug development," Crews said.
More information: "Catalytic in vivo protein knockdown by small-molecule PROTACs" Nature Chemical Biology (2015) DOI: 10.1038/nchembio.1858
Journal information: Nature Chemical Biology
Provided by Yale University