New electronic stent could provide feedback and therapy—then dissolve
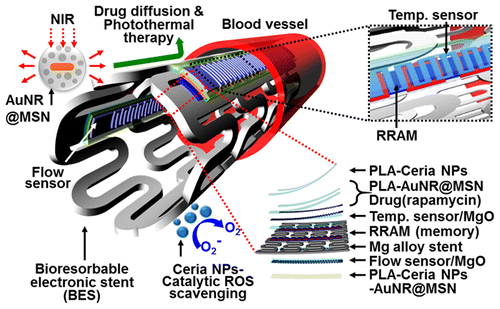
Every year, an estimated half-million Americans undergo surgery to have a stent prop open a coronary artery narrowed by plaque. But sometimes the mesh tubes get clogged. Scientists report in the journal ACS Nano a new kind of multi-tasking stent that could minimize the risks associated with the procedure. It can sense blood flow and temperature, store and transmit the information for analysis and can be absorbed by the body after it finishes its job.
Doctors have been implanting stents to unblock coronary arteries for 30 years. During that time, the devices have evolved from bare metal, mesh tubes to coated stents that can release drugs to prevent reclogging. But even these are associated with health risks. So researchers have been working on versions that the body can absorb to minimize the risk that a blood clot will form. And now Dae-Hyeong Kim, Seung Hong Choi, Taeghwan Hyeon and colleagues are taking that idea a step further.
The researchers developed and tested in animals a drug-releasing electronic stent that can provide diagnostic feedback by measuring blood flow, which slows when an artery starts narrowing. The device can also heat up on command to speed up drug delivery, and it can dissolve once it's no longer needed.
More information: Bioresorbable Electronic Stent Integrated with Therapeutic Nanoparticles for Endovascular Diseases Bioresorbable Electronic Stent Integrated with Therapeutic Nanoparticles for Endovascular Diseases, ACS Nano, Article ASAP. DOI: 10.1021/acsnano.5b00651
Abstract
Implantable endovascular devices such as bare metal, drug eluting, and bioresorbable stents have transformed interventional care by providing continuous structural and mechanical support to many peripheral, neural, and coronary arteries affected by blockage. Although effective in achieving immediate restoration of blood flow, the long-term re-endothelialization and inflammation induced by mechanical stents are difficult to diagnose or treat. Here we present nanomaterial designs and integration strategies for the bioresorbable electronic stent with drug-infused functionalized nanoparticles to enable flow sensing, temperature monitoring, data storage, wireless power/data transmission, inflammation suppression, localized drug delivery, and hyperthermia therapy. In vivo and ex vivo animal experiments as well as in vitro cell studies demonstrate the previously unrecognized potential for bioresorbable electronic implants coupled with bioinert therapeutic nanoparticles in the endovascular system.
Journal information: ACS Nano
Provided by American Chemical Society