How unwanted CDs and DVDs could help cut carbon emissions
Now that most consumers download and stream their movies and music, more and more CDs and DVDs will end up in landfills or be recycled. But soon these discarded discs could take on a different role: curbing the release of greenhouse gases. In the journal ACS Sustainable Chemistry & Engineering, scientists report a way to turn the discs into a material that can capture carbon dioxide, a key greenhouse gas, and other compounds.
Mietek Jaroniec and colleagues from Poland and the U.S. note that manufacturers typically use natural sources, such as coal and wood, to make activated carbon. The material is then incorporated in a wide range of applications from decaffeination to gas purification. More recently, scientists have been preparing activated carbon out of everyday plastic products. Jaroniec's team wanted to try this with optical discs, a fast-growing part of our waste stream.
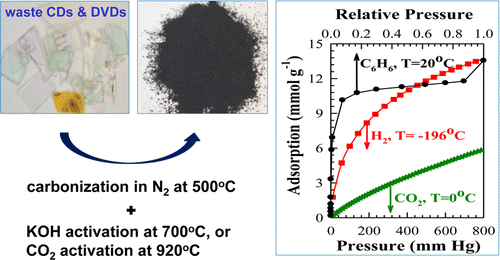
The researchers processed disc fragments into two kinds of activated carbon with high surface areas and large volumes of fine pore. These key characteristics allowed the materials to capture carbon dioxide. They also adsorbed hydrogen gas and benzene, a carcinogenic compound used in industrial processes. The researchers say that in addition to carbon capture applications, their materials could be used to separate volatile organic compounds and store hydrogen.
More information: Adsorption Properties of Activated Carbons Prepared from Waste CDs and DVDs, ACS Sustainable Chem. Eng., 2015, 3 (4), pp 733–742. DOI: 10.1021/acssuschemeng.5b00036
Abstract
Two sets of activated carbons have been prepared from waste CDs and DVDs by carbonization and subsequent activation with either KOH or CO2. The resulting activated carbons had specific surface area in the range of 500–2240 m2 g–1, total pore volume in the range of 0.18–1.36 cm3 g–1, volume of micropores and small mesopores (w < ∼2.9 nm) in the range of 0.17–1.25 cm3 g–1, and volume of small micropores (w < ∼1.2 nm) in the range of 0.14–0.71 cm3 g–1. Both KOH and CO2 activation resulted in 5–45-fold improvement in the structural properties, depending on the conditions used. The resulting carbons showed good adsorption properties toward carbon dioxide, hydrogen, and benzene. The best uptakes for these adsorptives were 5.8 mmol g–1 of CO2 at 0 °C and 800 mmHg, 3.3 mmol g–1 of CO2 at 25 °C and 850 mmHg, 13.9 mmol g–1 of H2 at –196 °C and 850 mmHg, and 15.4 mmol g–1 of C6H6 at 20 °C and saturation pressure. The excellent adsorption properties of the prepared carbons render them as potential adsorbents in CO2 capture and storage, VOCs adsorption/separation, and hydrogen storage.
Provided by American Chemical Society