February 18, 2015 feature
Red light goes green: Metal-free organic sensitizers portend significant advance in artificial photosynthesis
(Phys.org)—Photosynthesis – the ubiquitous yet remarkable process by which most plants, algae, and cyanobacteria convert light energy into chemical energy – provides the atmospheric oxygen and organic compounds fundamental to the evolution of life on Earth, and in so doing captures some six times more energy than humans consume while annually converting roughly 100 billion tons of carbon into biomass. In the effort to find alternatives to fossil fuels, the field of artificial photosynthesis – a chemical process that replicates natural photosynthesis – seeks to capture and store energy from visible light, including sunlight, in the chemical bonds of what is known as a solar fuel (for example, hydrogen, methane or methanol).
To be both effective and efficient, artificial photosynthesis requires tunable photosensitizers (light-absorbing molecules also referred to simply as sensitizers that mediate reactions to light) to capture, convert, and transfer visible light energy. To also be practical, these sensitizers must have a very specific absorption, excitation, oxidative, and stability profile; react strongly to red light; and use readily available materials. Recently, scientists at Pennsylvania State University, University Park and Arizona State University, Tempe designed and demonstrated metal-free organic photosensitizers that meet these criteria, thereby achieving photoelectrochemical water oxidation at a level comparable to sensitizers based on rare materials while being the first artificial molecules to oxidize water using only red light.
Prof. Thomas E. Mallouk discussed the paper that he, Dr. John R. Swierk and their co-authors published in Proceedings of the National Academy of Sciences. Describing the challenges of showing that metal-free organic photosensitizers are capable of driving photoelectrochemical water oxidation, Mallouk points out that while water splitting to hydrogen and oxygen is a thermodynamically straightforward reaction – it can be driven by light anywhere in the visible spectrum – a detailed thermodynamic analysis published in 1985 showed that light of wavelength shorter than 775 nm can in principle drive efficient solar water splitting1. "However," Mallouk tells Phys.org, "there are a number of practical constraints for a system in which light is absorbed by a molecule and the molecule then oxidizes water to make oxygen" Excited states do not last long, and there is a strong driving force for them to return to the ground state, usually by a series of thermodynamically downhill and rapid electron transfer steps."
In both natural and artificial photosynthetic systems, the light-absorbing molecule in its excited state transfers an electron to an acceptor, which in the research being discussed is an oxide semiconductor electrode. "The oxidized form of the molecule must then take an electron from water – usually via another molecule that catalyzes water oxidation – before the photoinjected electron comes back from the semiconductor and regenerates the ground state. The back electron transfer process is parasitic, in that it converts the energy of the excited state into heat." Moreover, Mallouk says, back electron transfer is typically fast – occurring in 100 microseconds or less – and so to oxidize water the oxidized dye molecule needs to work quickly, meaning that it has to be a fairly powerful oxidizer – at least 300 mV more positive in the electrochemical series than the oxygen/water couple.
"There are not many molecules that combine all the right properties," Mallouk notes, these being:
- strongly absorbing in the visible spectrum
- long-lived excited state that is energetically capable of rapidly injecting an electron into the conduction band of titanium dioxide (TiO2)
- highly oxidizing in its oxidized form
- chemically stable over many cycles of photoredox reactions (those that harness visible light energy to accelerate a chemical reaction via a single-electron transfer)
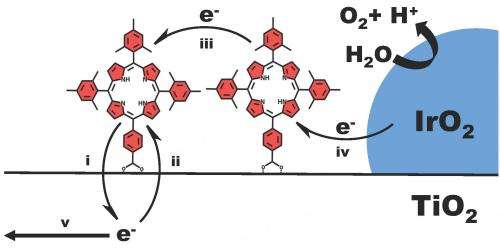
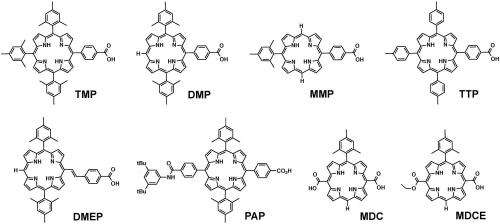
"To date," he adds, "the field has been dominated by studies of ruthenium- and iridium-containing coordination compounds that combine these properties – but our paper explores the idea of using porphyrins in place of ruthenium and iridium complexes in a water-splitting dye-sensitized photoelectrochemical cell." Porphyrins (macrocyclic organic compounds (that is, those with a ring structure consisting of 12 or more atoms) structurally related to the pigments in chlorophyll and hemoglobin are strongly absorbing. Moreover, with the right substituents (atoms or group of atoms taking the place of another atom or group or occupying a specified position in a molecule), porphyrins have the photoredox properties needed to drive water oxidation in a dye-sensitized electrode – and unlike ruthenium and iridium, are neither rare nor expensive. To accomplish this, the scientists had to synthesize the dye molecules, incorporate them into the cell with appropriate catalyst particles, and quantify the oxygen generated at the illuminated photoelectrodes.
The researchers also demonstrated that under broadband illumination, these metal-free organic photosensitizers exhibit activity comparable to that of ruthenium-containing photosensitizers. "The activity is assessed by measuring the photocurrent and oxygen evolution yield as a function of the applied bias potential," Mallouk explains. "Earlier studies had already established the 2:1 H2/O2 stoichiometry of water splitting in this kind of cell, where hydrogen is made by reduction of water at a dark platinum counter electrode." (Stoichiometry is the relationship between the relative quantities of substances taking part in a reaction or forming a compound, typically a ratio of whole integers.)
Another important finding presented in the paper was that this is the first molecular photosensitizer, outside of natural photosynthesis, that can drive water oxidation utilizing only red light. (Previous research based on ruthenium split water – but only using blue light.) Mallouk emphasizes that since porphyrins absorb strongly between the Soret band and the Q-bands (400-450 nm and 500-670 nm, respectively), it was important to show that the system worked under red light (wavelengths longer than 590 nm) – that is, so that absorption was occurring only in the Q-band region – because most of the photon flux in sunlight is in the red and near-infrared part of the spectrum. "Previously" he points out, "only metal-containing sensitizers that absorb in the blue at wavelengths shorter than 500 nm had been used in this kind of water splitting cell."
A significant challenge the Arizona State part of the team faced was to design and synthesize porphyrin molecules that were sufficiently oxidizing to drive the rapid oxidation of water, as well as sufficiently reducing in their excited state to rapidly inject an electron into the titanium dioxide conduction band. "We tuned the oxidizing power of the porphyrins by placing appropriate substituents on the porphyrin ring," Mallouk recounts. "In addition, the porphyrins needed to be designed with specific functional groups that would allow attachment to the oxide electrode surface." To that end, the Penn State part of the team had to determine the right conditions for adsorbing these molecules on the electrode surface, which was made easier by their already having worked out the testing conditions and the methods for determining electron transfer rate constants from earlier studies with other dye molecules.
The paper also notes that while the open-circuit photovoltages and integrated photocurrents are quite similar for most of the free-base porphyrin sensitizers, the scientists' deeper analysis of the spectral and electrochemical properties of the sensitizers reveals significant differences. "We found that changing the substituents on the porphyrin ring had a relatively small effect on the energy of the excited state, which correlates with the difference between the excited and ground state oxidation potentials," Mallouk explains. "However, changing the substituents did significantly shift these potentials relative to the oxygen/water potential." For example, with electron-withdrawing substituents the oxidized form of the molecule was a more powerful oxidant, but the excited state was a weaker reductant. "There's a tradeoff between these properties since one would like both electron injection from the excited state and oxidation of water from the oxidized form of the dye to be fast processes – and the rates of these processes depend on and are sensitive to the electrochemical driving force for electron transfer."
In the paper, the scientists discuss their ideas on developing a kinetic picture to enable rational design strategies for improving the system. Previously, they had developed2 a model for dye-sensitized water splitting photoelectrochemical cells that incorporates the kinetics of all the electron transfer reactions in the system. "These processes," Mallouk tells Phys.org, "include electron injection from the excited state molecule into the TiO2 semiconductor, electron transfer from the water oxidation catalyst to the oxidized dye, electron transport in the TiO2 electrode, electron hopping between sensitizer molecules, and two parasitic recombination processes – that is, back electron transfer from TiO2 to the oxidized dye, and electron scavenging by the water oxidation catalyst. While the recombination processes are winning the battle, so the overall quantum yield of water splitting is low, we know that all the electron transfer rates depend strongly on the distance between redox partners – so we hope to slow down the parasitic reactions using core-shell electrode structures, a strategy already demonstrated with ruthenium-containing sensitizers by our team and by Prof. Thomas J. Meyer's group at the University of North Carolina at Chapel Hill. We're also investigating molecular catalysts in order to shorten the electron hopping pathway from the dye to the catalyst." What's critical is that each of these design ideas can be tested by measuring the kinetic rate constants, which the scientists obtain from the time-dependent photocurrent and photovoltage in the cell.
A central point made in the paper is that the utilization of red light to drive water splitting represents a significant step forward for molecular photoelectrochemical water-splitting systems. "Yes, it's an important step," Mallouk explains, "because red light accounts for most of the energy in the solar spectrum. In addition, there are other macrocyclic compounds that are structurally related to porphyrins that while being a bit more challenging to synthesize are much stronger red light absorbers. We hope to investigate those molecules in the future."
In addition to developing core-shell structures and molecular catalysts, as discussed above, the scientists are creating sensitizer molecules that should absorb more strongly in the red visible spectrum. Moreover, the Arizona State team has made linked sensitizer-mediator molecules that speed up electron transfer between the catalyst and the sensitizer molecule. "We're also working on a molecular photocathode assembly that should develop sufficient photovoltage to generate hydrogen without applying a bias voltage to the cell," Mallouk adds. "Together with the photoanode assembly, this will make a photoelectrochemical 'Z-scheme' in which two photons are absorbed – one at the anode, the other at the cathode – for each electron transferred in the cell." The so-called Z‐scheme typically refers to oxidation/reduction processes in reactions to light during natural photosynthesis.
"As part of this project," Mallouk concludes, "we and other groups have begun to develop membranes for use in water splitting solar cells3,4,5. "We believe that the membrane results could be broadly applicable to electrolyzers and related technologies for solar fuels, including systems that reduce CO2 to carbon-containing fuels."
More information: Metal-free organic sensitizers for use in water-splitting dye-sensitized photoelectrochemical cells, Proceedings of the National Academy of Sciences Published online before print January 12, 2015, doi:10.1073/pnas.1414901112
Related:
1Limiting and realizable efficiencies of solar photolysis of water, Nature (1985) 316: 495-500, doi:10.1038/316495a0
2Effects of electron trapping and protonation on the efficiency of water-splitting dye-sensitized solar cells, Journal of the American Chemical Society (2014) 136:10974-10982, doi:10.1021/ja5040705
3Resistance and polarization losses in aqueous buffer–membrane electrolytes for water-splitting photoelectrochemical cells, Energy & Environmental Science (2012) 5:7582-7589, doi:10.1039/C2EE03422K
4Assessing the utility of bipolar membranes for use in photoelectrochemical water-splitting cells, Chemistry & Sustainability, Energy & Materials, (2014) 7:3017–3020, doi:10.1002/cssc.201402535
5Use of bipolar membranes for maintaining steady-state pH gradients in membrane-supported, solar-driven water splitting, Chemistry & Sustainability, Energy & Materials, (2014) 7:3021-3027, DOI: 10.1002/cssc.201402288
© 2015 Phys.org