Muons help understand mechanism behind hydrogen storage
It is ever more necessary to find alternative ways to store energy. Energy storage is required when energy is supplied intermittently, as for instance for wind power, or for mobile applications like cars. Hydrogen is a promising solution due to the large amount of energy produced by its clean reaction with oxygen.
Muon spin relaxation reveals promising storage materials
To be stored, hydrogen needs to bind with some material. Li6C60 and Na10C60 fullerides are promising hydrogen storage systems as they are light, easily processed by industry, and able to reversibly absorb hydrogen. It is known that they absorb up to 5 mass % of hydrogen but the mechanisms driving the absorption were not well-understood.
Scientists from the University of Parma, Uni. of Pavia, and the Institute Laue-Langevin have used muon spin relaxation to investigate hydrogenation on the Li6C60 and Na10C60. Muons capture electrons to form muonium, a light isotope of hydrogen. Thanks to this powerful technique it is possible to observe how muonium behaves and thus understand how hydrogen interacts with matter. The group has received NMI3 support to conduct experiments at the ISIS Pulsed Muon Facility.
On the way to more efficient hydrogen storage
The scientists learned that unlike many other fullerides, in Li6C60 and Na10C60, metal atoms group themselves together, which fosters the formation of muonium. This makes them perfect test-materials for this study.
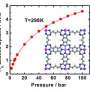
Surprisingly, the results suggest that the hydrogenation process is more effective at very low temperatures, below -150 ⁰C, when hydrogen is atomic. So why does hydrogenation seem to perform better at high temperatures? Hydrogen (H2) first needs to dissociate into H, which will then be absorbed by the material used for storage via a chemical reaction. It is in fact the dissociation of hydrogen that requires high temperatures.
Further research on these dynamics could reveal ways to decrease the temperature required to store hydrogen and adapt these materials accordingly, making hydrogen storage more efficient and cheaper.
Provided by CORDIS