Skin patch could replace the syringe for disease diagnosis
Drawing blood and testing it is standard practice for many medical diagnostics. As a less painful alternative, scientists are developing skin patches that could one day replace the syringe. In the ACS journal Analytical Chemistry, one team reports they have designed and successfully tested, for the first time, a small skin patch that detected malaria proteins in live mice. It could someday be adapted for use in humans to diagnose other diseases, too.
Simon R. Corrie and colleagues note that while blood is rich with molecular clues that tell a story about a person's health, withdrawing it is often painful. It also requires trained personnel and expensive lab equipment and facilities for analysis. These factors don't lend themselves to patient compliance or accessibility in resource-limited places. Scientists have been trying to address these hurdles by developing diagnostic patches that are covered on one side with thousands of microscopic, hollow needles that can sample fluid in the skin. But so far, these devices have only been able to test for one compound at a time. However, many diseases can be diagnosed more reliably by detecting multiple biomarkers. Corrie's team wanted to design a new patch that could meet this need.
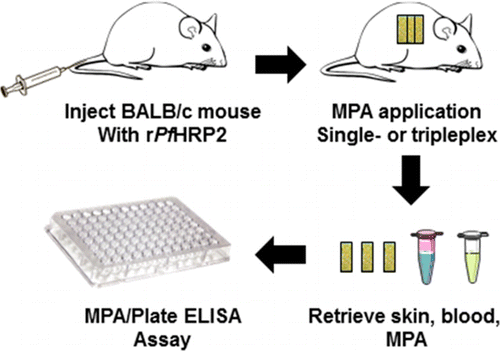
The researchers optimized their device so it could capture two biomarkers for the malaria parasite, Plasmodium falciparum, which kills more than 1 million people every year. To test it, they injected malaria proteins into the bloodstream of live mice and applied the patch to their skin. The patch successfully captured the proteins in the skin tissue. Such devices, they conclude, could be used in the future to diagnose malaria and other infectious diseases in a less painful way.
More information: "Capture of the Circulating Plasmodium falciparum Biomarker HRP2 in a Multiplexed Format, via a Wearable Skin Patch" Anal. Chem., 2014, 86 (20), pp 10474–10483. DOI: 10.1021/ac5031682
Abstract
Herein we demonstrate the use of a wearable device that can selectively capture two distinct circulating protein biomarkers (recombinant P. falciparum rPfHRP2 and total IgG) from the intradermal fluid of live mice in situ, for subsequent detection in vitro. The device comprises a microprojection array that, when applied to the skin, penetrates the outer skin layers to interface directly with intradermal fluid. Because of the complexity of the biological fluid being sampled, we investigated the effects of solution conditions on the attachment of capture antibodies, to optimize the assay detection limit both in vitro and on live mice. For detection of the target antigen diluted in 20% serum, immobilization conditions favoring high antibody surface density (low pH, low ionic strength) resulted in 100-fold greater sensitivity in comparison to standard conditions, yielding a detection limit equivalent to the plate enzyme-linked immunosorbent assay (ELISA). We also show that blocking the device surface to reduce nonspecific adsorption of target analyte and host proteins does not significantly change sensitivity. After injecting mice with rPfHRP2 via the tail vein, we compared analyte levels in both plasma and skin biopsies (cross-sectional area same as the microprojection array), observing that skin samples contained the equivalent of ∼8 μL of analyte-containing plasma. We then applied the arrays to mice, showing that surfaces coated with a high density of antibodies captured a significant amount of the rPfHRP2 target while the standard surface showed no capture in comparison to the negative control. Next, we applied a triplex device to both control and rPfHRP2-treated mice, simultaneously capturing rPfHRP2 and total IgG (as a positive control for skin penetration) in comparison to a negative control device. We conclude that such devices can be used to capture clinically relevant, circulating protein biomarkers of infectious disease via the skin, with potential applications as a minimally invasive and lab-free biomarker detection platform.
Journal information: Analytical Chemistry
Provided by American Chemical Society