Developing the battery of the future

The search for the next generation of batteries has led researchers at the Canadian Light Source synchrotron to try new methods and materials that could lead to the development of safer, cheaper, more powerful, and longer-lasting power sources, to be used in almost everything, from vehicles to phones.
"Typically, battery research involves cooking chemicals together to create new materials," said Dr. Jigang Zhou, CLS industrial staff scientist. "The performance of these materials is measured by testing the current, voltage, charge time, and number of charge cycles the materials can take.
"Essentially, you take a stab in the dark and see how good your aim was," said Zhou.
But there are problems with this method, and researchers are still uncertain why some materials work better than others. So Zhou and other researchers are using the synchrotron to examine these materials in a whole new way.
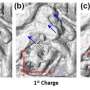
If you think of a typical AA battery, the kind you would find in most TV remotes, there is a positive and a negative end called an electrode. Although a number of materials can be used for these electrodes, Zhou and his team are using a novel lithium-nickel-maganese-oxide (LMNO) material on the positive electrode that could provide batteries with significantly higher voltage.
While the higher voltage of this material can offer a real advantage, it tends to dry out the electrolyte – a liquid necessary for batteries to work properly. Understanding the role that each element in LMNO plays is critical to furthering his research into why the electrolyte dries up.
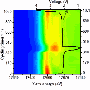
Synchrotron X-rays allows for the visualization of the LMNO is such fine detail that he can identify where the material is breaking down the electrolyte liquid, and determine what is happening and how to prevent the breakdown in those places.
For the negative electrode however, silicon is a promising and cheap material that researchers are also having success with.
"Silicon offers the potential for a higher capacity battery that could hold more charge storage per gram when compared to conventional batteries," he said. "Such a battery could work longer after a single charge."
Zhou stated "the capacity for silicon is 10 times greater than current negative electrode materials." However, this material comes with its own challenges. When silicon is used in a battery, its volume changes more greatly between when the battery is fully charged and when it has used up its charge. These changes in volume during charging cycles cause the battery to break down over time, so researchers are working on ways to make silicon more stable so it can be used commercially.
Zhou believes that since there are currently no batteries on the market using silicon-LMNO electrodes, research into perfecting these materials could lead to new, better batteries.
Provided by Canadian Light Source