Toward making lithium-sulfur batteries a commercial reality for a bigger energy punch

A fevered search for the next great high-energy, rechargeable battery technology is on. Scientists are now reporting they have overcome key obstacles toward making lithium-sulfur (Li-S) batteries, which have the potential to leave today's lithium-ion technology in the dust. Their study appears in the ACS journal Nano Letters.
Xingcheng Xiao, Weidong Zhou, Mei Cai and their colleagues point out that the capabilities of lithium-ion batteries, which power many of our consumer electronics, as well as electric vehicles, have largely plateaued. Scientists have been pursuing a number of new battery technologies to topple today's standard. One heavy focus has been on a key battery component that is currently made out of a metal oxide. Some researchers have been trying to replace the metal oxide with cheaper and lighter sulfur, to make Li-S batteries. In theory, this could allow batteries to pack five to eight times the energy of existing technology. One of the main problems with this approach, however, is that Li-S compounds escape from where they're supposed to be, which causes the battery to lose charge quickly. The team set out to find a way to contain the errant compounds.
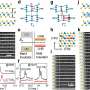
To solve this problem, the researchers made tiny, hollow shells out of carbon, which is conductive. They then coated them with a polymer to help confine the Li-S compounds inside. When tested, the structures kept up a high-energy storage capacity (630 mAh/g versus less than 200 mAh/g of lithium-ion batteries) over 600 cycles of fast charging and discharging. "These results provide promising insights and novel concepts for future sulfur-based batteries," the researchers conclude.
More information: "Polydopamine-Coated, Nitrogen-Doped, Hollow Carbon-Sulfur Double-Layered Core-Shell Structure for Improving Lithium-Sulfur Batteries" Nano Lett., 2014, 14 (9), pp 5250–5256. DOI: 10.1021/nl502238b
Abstract
To better confine the sulfur/polysulfides in the electrode of lithium–sulfur (Li/S) batteries and improve the cycling stability, we developed a double-layered core–shell structure of polymer-coated carbon–sulfur. Carbon–sulfur was first prepared through the impregnation of sulfur into hollow carbon spheres under heat treatment, followed by a coating polymerization to give a double-layered core–shell structure. From the study of scanning transmission electron microscopy (STEM) images, we demonstrated that the sulfur not only successfully penetrated through the porous carbon shell but also aggregated along the inner wall of the carbon shell, which, for the first time, provided visible and convincing evidence that sulfur preferred diffusing into the hollow carbon rather than aggregating in/on the porous wall of the carbon. Taking advantage of this structure, a stable capacity of 900 mA h g–1 at 0.2 C after 150 cycles and 630 mA h g–1 at 0.6 C after 600 cycles could be obtained in Li/S batteries. We also demonstrated the feasibility of full cells using the sulfur electrodes to couple with the silicon film electrodes, which exhibited significantly improved cycling stability and efficiency. The remarkable electrochemical performance could be attributed to the desirable confinement of sulfur through the unique double-layered core–shell architectures.
Journal information: Nano Letters
Provided by American Chemical Society