Finding the genetic culprits that drive antibiotic resistance
Researchers have developed a powerful new tool to identify genetic changes in disease-causing bacteria that are responsible for antibiotic resistance. The results from this technique could be used in clinics within the next decade to decide on the most effective treatments for diseases such as pneumonia and meningitis.
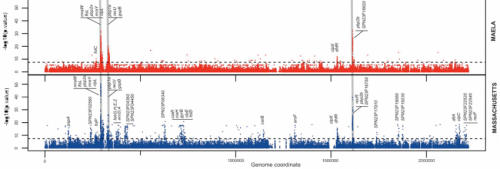
The team looked at the genome of Streptococcus pneumoniae, a bacterial species that causes 1.6 million deaths worldwide each year. In the most detailed research of its kind, scientists used a genome-wide association study (GWAS) to locate single-letter changes in the DNA code of the bacterium, which enable it to evade antibiotic treatment.
While GWAS has been used for a decade to identify gene function in humans, it was thought to be impossible to use the technique on bacterial DNA until now.
"The results of this research are very interesting," says Claire Chewapreecha, first author from the Wellcome Trust Sanger Institute. "For the first time, we are able to see, at large scale, causative variants that allow bacteria such as Streptococcus pneumoniae to resist our efforts to treat and control it.
"We can begin to see how this might help us to develop more effective treatment strategies in the near future."
GWAS studies search through the genome for locations where single DNA changes are associated with properties of the organism, like antibiotic resistance. For this to work, it is essential that a genetic exchange process called recombination has occurred. This is where two DNA sequences combine, exchanging genetic data, and shuffling combinations of single changes. Because recombination, which is very common in humans, is rare in bacteria, researchers have previously been unable to locate the individual changes in the sequence of bases that make up a gene. Until now it has only been possible to locate the general area where change has occurred in so-called mosaic genes comprised of genetic data from multiple strains of bacteria.
To overcome this hurdle, researchers used the richest available set of data for S. pneumoniae, which was collected by Dr Claudia Turner, a Research Paediatrician, and Dr Paul Turner, a Clinical Microbiologist, at the Mahidol Oxford Tropical Medicine Research Unit. The dataset of over 3,000 samples of Streptococcus pneumoniae isolated from almost 1,000 infants and mothers at a refugee camp on the border between Myanmar and Thailand provided enough recombination events to give researchers precise data about the locations of changes that cause resistance. A second set of several hundred isolates was collected from Massachusetts as part of a project to assess the impact of the vaccine against S. pneumoniae introduced in the USA in 2001.
"In this study we've shown that this powerful genetic tool, which has transformed our understanding of human genetics, can be applied to bacteria," says Professor Stephen Bentley, a senior author from the Sanger Institute. "This opens up new avenues of research into antibiotic resistance, transmission and virulence that were previously thought impossible in bacterial genomics."
The next phase of research will involve fine tuning the technique to be able to identify genes in bacteria that make strains more virulent and genes that enable transmission of a bacterial strains between hosts. As genetic sequencing moves into clinics, this detailed understanding will inform control and treatment strategies.
"Uncovering all the single-letter differences underlying resistance will be essential for future use of genome sequencing to predict antibiotic sensitivity in clinical microbiology," says Professor Julian Parkhill. "GWAS will enable this by allowing us to pinpoint the location of the real genetic culprits rather than our current mapping that gets us only to the right general area."
More information: Comprehensive Identification of Single Nucleotide Polymophisms Associated with Beta-lactam Resistance within Pneumococcal Mosaic Genes. Chewapreecha, C, et al. PLOS Genetics 2014 DOI: 10.1371/journal.pgen.1004547 . dx.doi.org/10.1371%2Fjournal.pgen.1004547
Provided by Wellcome Trust Sanger Institute