The large wandering spider bites forever

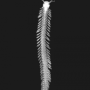
The fangs of the large wandering spider (Cupiennius salei) are incisively shaped, right to the tip. The spider uses its curved fang to grasp its prey, punctures its chitinous armour and injects venom into the victim. As a research team from the Max Planck Institute of Colloids and Interfaces in Potsdam-Golm and the University of Vienna have established, the shape and structure of the fangs have evolved extremely well to perform its lethal task multiple times over the spider's lifetime, as reliably as possible and with the minimum amount of damage. For the first time, the researchers analysed in detail the correlation between the structure and the mechanical properties of the fang and compared it with other potential configurations. This also provided starting points for investigating how bio-inspired injection equipment in medicine and technology can be improved.
Evolution operates a global laboratory on earth. While it cannot call on as many materials as engineers can, it makes up for this disadvantage with an incredible variety of material combinations, structures and shapes that have emerged over the millennia. The venomous fangs of spiders provide convincing proof of this. The armour of insects eaten by spiders consists of chitin and proteins, the same material of which the fangs are built. "However, the architecture of the fang is adapted in such a way that it can penetrate the armour, suffering little or no damage," says Yael Politi, who, together with Benny Bar-On and Peter Fratzl from the Max Planck Institute of Colloids and Interfaces and Friedrich G. Barth from the University of Vienna, studied the biophysics of the fang with which the large wandering spider (Cupiennius salei) lays low its victims.
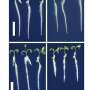
The researchers first examined the shape of the fang: a conical structure, curved in shape, with a venom canal running through it, whose wall is thicker at the base than at the tip. Using a computational model, the scientists tested the properties of this architecture and its behaviour under associated mechanical loads. They varied the geometry of the model fang, creating a variety of structures: a curved, needle-shaped fang, a fang structure almost identical to that found in nature and a blunter cone shape with a wider base containing significantly more material than the natural fang. "As our simulations show, the shape makes the venomous fang very stiff with a relatively small amount of material," says Benny Bar-On. "The fang therefore bends with higher loads compared to the behaviour of a curved needle-like structure, for example."
In the case of damage, the spider does not lose the entire fang
The natural fang is subject to the maximum stress at the tip if the spider circles in on its prey. In contrast, with a needle shape, the greatest stress occurs at the base. If the fangs were needle shaped, a break would cause the spider to lose its fangs. If, on the other hand, the natural fang is damaged, the worst that can happen is that it loses its tip; it can still use the fang to hold the prey with the remaining stump. "With a gradient, namely a change in the wall's thickness, nature has achieved greater resilience," says Benny Bar-On.
Similarly, the internal structure of the venomous fang is well adapted, or more precisely, the arrangement of the chitin fibres, which are embedded in proteins. The outer layer of the fang consists of alternating chitin layers in which the fibres are arranged in parallel. Like a plywood board, however, the direction of the fibres differs from one layer to the next. In a middle layer, on the other hand, the fibres all run parallel to the direction in which the fang strikes the armour of the prey in a classic bite. An inner layer of the fang, which surrounds the venom canal, is again structured like a sheet of plywood that is made from layers of chitin that rotate against one another.
The sandwich structure supports the fang against various loads.
"The fang structure can absorb the actual loads effectively," says Benny Bar-On. "It helps the fang to withstand the pressure in the event of a frontal impact, but can also help it to resist shear stresses if the spider does not achieve the perfect bite and the fang is twisted." The researchers again realised this through simulations in which they varied the layer thickness virtually in different ways. As expected, the fang whose chitin fibres run mainly parallel to the optimum direction of movement proved to be particularly resilient to frontal loads. However, if a spider with this kind of fang is somewhat careless when it bites its prey and the fang is twisted slightly when it impacts its prey, the injection needle would quite easily break. And it would most likely break at the base, which would mean that the spider would in turn lose its entire weapon. This is prevented not only by the sandwich structure with various fibril orientations, but also by variations in the fiber architecture of the fang from tip to base. At the tip, a relatively large number of fibres, running in parallel to the optimum bite movement, absorb the force of the impact. At the base, a larger proportion of fibres in the plywood design stabilises the fang against shear stresses. Thus, evolution has resolved this optimisation problem as well as possible.
The model simulations could explain the evolved structure.
"With the help of to the model simulations, we can now suggest explanations for why the evolution of the spider's fang has resulted in the design that we find in nature," says Benny Bar-On. In other words, they can suggest why the fang is conical in shape and is not needle-shaped, like a mosquito proboscis.
"In our study, we systematically examined the correlation between the structure and the biomechanical properties of a biological structure at different levels and focused particularly on the interaction between the various hierarchical levels," explains Yael Politi. The researchers also expect interesting insights when other systems from nature's archive are analysed in this way. In particular, they expect to learn a lot from looking at structures which, like the spider's fang, feature gradients or local variations. This applies, for example, to the foot that attaches a mussel to the ocean bed or the beak of a cuttlefish.
Should the analysis of such an organism show that evolution – still – falls short of the optimum, this may not help the organism in question. For materials scientists and engineers, however, such a finding could be very valuable indeed. It would mean that they could avoid nature's inadequacies when they design a device inspired by biology. Such model simulations would also enable them to adapt nature's developments to specific technical requirements with relatively little effort. Analysing the spider's fang could therefore have a practical outcome, such as on the design of injection valves in motors or injection or suction devices in medicine.
More information: Benny Bar-On, Friedrich G. Barth, Peter Fratzl & Yael Politi. "Multiscale structural gradients enhance the biomechanical functionality of the spider fang." Nature Communications, 27 May 2014; DOI: 10.1038/ncomms4894
Journal information: Nature Communications
Provided by Max Planck Society