April 11, 2014 report
Research team develops method to strengthen elastomers
(Phys.org) —A team of researchers working in France has found a way to toughen elastomers—a class of rubbers. In their paper published in the journal Science, the team describes how they used a technique similar to that used to strengthen hydrogels to make elastomers stronger. Jian Ping Gong offers a Perspective piece on the work in the same issue of the journal.
Elastomers are used in a wide variety of applications, primarily as shock absorbers—they can generally be easily stretched, but tend to break if a notch develops. Scientists would like to improve the strength of elastomers because it would allow for their use in more applications and for better results in applications in which they are already used. In this new effort, the researchers looked to a technique that has proven to be a good method of strengthening hydrogels—combining two different network materials.
Hydrogels, as their name implies, are made mostly of water, they resemble organic tissue in many respects which is why research is ongoing to find a way to use them in biological applications. Prior research has shown that the addition of a second network material can cause an increase in their strength—in essence the added strength comes from the mesh that is created, offering increased resistance to cracking while still maintaining pliability. Until now, however, applying the same technique to elastomers has proven to be challenging because a secondary network material that would offer contrasting properties was not known. The second is the tendency of non-water based materials to separate because they don't contain solvents. The team in France has found a solution to both these problems by employing a sequential polymerization technique.
To create an elastomer with added strength, the team started with a traditional network material. Instead of simply introducing a second network material, the researchers used monomers of the first material, causing it to swell—the monomers were then polymerized causing the completion of a double-network structure. The end result is an elastomer that is stronger than those that have been made before, yet is still able to be stretched. Interestingly, the process can be repeated again to create a triple-network structure elastomer as well.
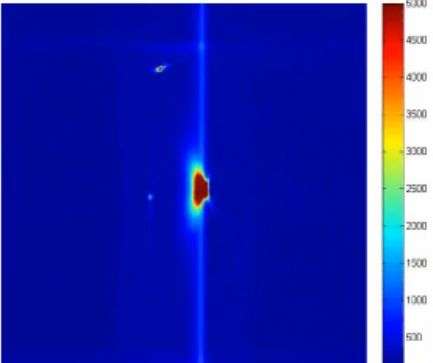
To help test their new material, the researchers also added chemoluminescent molecules into the mix which caused the emittance of light when the material cracked. As Gong notes, the new technique is likely to stoke interest by other researchers, leading perhaps, to a new class of products.
More information: Toughening Elastomers with Sacrificial Bonds and Watching Them Break, Science 11 April 2014: Vol. 344 no. 6180 pp. 186-189. DOI: 10.1126/science.1248494
ABSTRACT
Elastomers are widely used because of their large-strain reversible deformability. Most unfilled elastomers suffer from a poor mechanical strength, which limits their use. Using sacrificial bonds, we show how brittle, unfilled elastomers can be strongly reinforced in stiffness and toughness (up to 4 megapascals and 9 kilojoules per square meter) by introducing a variable proportion of isotropically prestretched chains that can break and dissipate energy before the material fails. Chemoluminescent cross-linking molecules, which emit light as they break, map in real time where and when many of these internal bonds break ahead of a propagating crack. The simple methodology that we use to introduce sacrificial bonds, combined with the mapping of where bonds break, has the potential to stimulate the development of new classes of unfilled tough elastomers and better molecular models of the fracture of soft materials.
Journal information: Science
© 2014 Phys.org