April 28, 2014 report
Researchers able to watch phase transition in 2D semiconductors using STEM
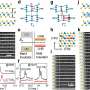
(Phys.org) —A team of researchers with members from Japan, Taiwan and Switzerland has discovered that it is possible to watch a phase transition occur in a 2D semiconducting material using a scanning transmission electron microscope (STEM). In their paper published in the journal Nature Nanotechnolgy describing their research and results, the team outlines how they used the microscope to watch as a sample of the direct bandgap semiconductor molybdenum sulphide underwent a phase shift.
An ability to phase shift between metallic capabilities and a semiconductor is an important feature of a material—one that scientists would like to better understand. Up till now however, researchers had to infer some of what occurs when a material undergoes a phase shift, because they couldn't actually see it as it was happening. In this new effort, the researchers show that it is possible to directly watch a phase shift by doing so with a sample of molybdenum sulphide. In so doing, they have discovered that atom-by-atom movements are part of the shift, rather than complete shifts by a collective. The researchers suggest their observations hint at the prospect of creating layered 2D semiconductors "in-layer" rather than as a series of steps where one material is layered over another. That would allow for creating structures with atomic scale precision.
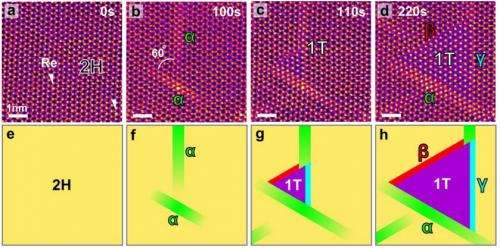
Molybdenum sulphide is polymorphic—it can function as either a metal or a semiconductor, depending on how much heat is present. Even better the two phases can be made to interconvert using intralayer atomic plane gliding, (a transversal displacement of one of the materials across the other) though it had never been seen actually doing so. As part of their research, the team performed in situ plane gliding while watching using the STEM, giving them an unprecedented view of what actually occurs as such phase shifting happens. The phase shift with the molybdenum sulphide sample occurred due to the heat exerted by the STEM itself. They suggest such a technique could also be used to induce phase shifting in other 2D materials.
The researchers also report that they have already used what they have learned to create several prototype nanodevices—one of which performs the functions of a Schottky diode.
More information: Atomic mechanism of the semiconducting-to-metallic phase transition in single-layered MoS2, Nature Nanotechnology (2014) DOI: 10.1038/nnano.2014.64 . On Arxiv: arxiv.org/ftp/arxiv/papers/1310/1310.2363.pdf
Abstract
Phase transitions can be used to alter the properties of a material without adding any additional atoms and are therefore of significant technological value. In a solid, phase transitions involve collective atomic displacements, but such atomic processes have so far only been investigated using macroscopic approaches. Here, we show that in situ scanning transmission electron microscopy can be used to follow the structural transformation between semiconducting (2H) and metallic (1T) phases in single-layered MoS2, with atomic resolution. The 2H/1T phase transition involves gliding atomic planes of sulphur and/or molybdenum and requires an intermediate phase (α-phase) as a precursor. The migration of two kinds of boundaries (β- and γ-boundaries) is also found to be responsible for the growth of the second phase. Furthermore, we show that areas of the 1T phase can be controllably grown in a layer of the 2H phase using an electron beam.
via Nanotechweb
Journal information: Nature Nanotechnology
© 2014 Phys.org