Nanoscale freezing leads to better imaging
It's an odd twist. For scientists to determine if a cell is functioning properly, they must destroy it.
This is what happens in X-ray fluorescence microscopy when biological specimens are exposed to ionizing radiation, which provides images with a level of detail that conventional microscopes just can't match. This exposure can change what is being imaged in profound ways, possibly giving false accounts of how the cell actually works.
To address this issue, researchers at the U.S. Department of Energy's (DOE) Argonne National Laboratory created a new probe that freezes cells to "see" at greater detail without damaging the sample.
The issue boils down to preparation.
Traditional X-ray methods look at cells that have either been immersed in water or dehydrated, like astronaut food. For wet specimens at room temperature, the radiation can break the bonds linking molecules together and cause them to scatter, changing the sample's structure.
For dehydrated specimens, potassium and other diffusible ions are washed away during chemical fixation, which kills the cell and loosens the cell membrane, allowing ions to escape. Moreover, when the sample is dehydrated, the cell can shrink, distort or even collapse.
"Imagine a ball. When you dry it, you make it flat," says Si Chen, principle author of the study. "It changes the structure of the sample and also the distribution of the trace elements that we are looking for."
To address this issue, Argonne researchers developed a hard X-ray fluorescence nanoprobe called the Bionanoprobe, which makes three-dimensional images that map out the locations of trace elements, like iron or potassium, in frozen biological samples.
"We don't want to dry the sample; we want to keep it hydrated," says Chen. "We plunge the sample into liquid ethane at very high speeds and then look at the frozen sample directly."
Rapidly cooling biological specimens to temperatures of -260°F preserves the natural state of a cell's organelles and trace elements while retaining the water in the sample.
Housed at an undulator beamline at sector 21 of Argonne's Advanced Photon Source, the Bionanoprobe features a vacuum chamber that eliminates frosting and convective heating and automatically acquires tomographic (sectioned images) data sets. Sector 21 is sponsored by a consortium of several universities and a research institute known collectively as the Life Sciences Collaborative Action Team.
The Bionanoprobe can also produce extremely high-resolution images at the smallest scales—below 100 nanometers. Compare that to a typical human hair, which is 80,000 to 100,000 nanometers wide. Chen uses X-ray optics called zone plates to focus the X-ray beam down to a miniscule small spot. A simple scan produces an image with a full fluorescent spectrum for each scanning step.
Recent tests have been encouraging. One team of researchers successfully acquired differential phase contrast and X-ray fluorescence images simultaneously by raster scanning of a green algae. The former gave researchers some of the algae's ultrastructure, and using the latter, they were able to show evenly distributed potassium and patterned distributions of zinc and iron.
"We can see the trace element distribution, but with biological samples, the contrast from the structure is typically very low," says Chen. "Phase contrast imaging highlights the structural details."
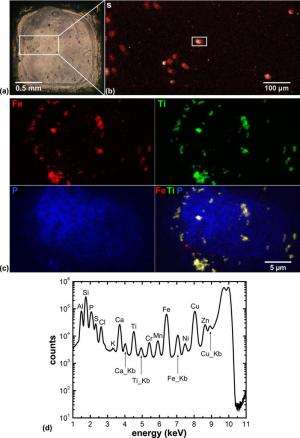
Another study made X-ray fluorescence images of an immortal cervical cancer cell line called HeLa cells. The samples were plunge-frozen, chemically fixed and then treated with an iron oxide core in a titanium dioxide shell nanocomposite, which allowed researchers to determine if the nanocomposites actually made it into the cell nucleus.
Dr. Gale Woloschak, professor at Northwestern University's Feinberg School of Medicine conducted the study. She created nanoparticles that target and kill cancer cells, but when the researchers wanted to see where the nanoparticles actually wound up in the cell, they ran into trouble with traditional X-ray methods.
"This is the problem," says Woloschak. "If you think of how two-dimensional X-ray imaging works, X-rays penetrate through the entire cell, so it's hard to determine whether the nanoparticles are above, below or inside the nucleus. What the Bionanoprobe does is give us a three-dimensional image—we could actually see that the nanoparticles were imbedded in the nucleus."
More information: "The Bionanoprobe: hard X-ray fluorescence nanoprobe with cryogenic capabilities." Chen S, Deng J, Yuan Y, et al. J Synchrotron Radiat. 2014 Jan;21(Pt 1):66-75. DOI: 10.1107/S1600577513029676.
Provided by Argonne National Laboratory