New method to create monomodal, mesoporous metal oxides

A team of UConn chemists has discovered a new way of making a class of porous materials that allows for greater manufacturing controls and has significantly broader applications than the longtime industry standard.
The process, more than three years in the making and outlined in the December 2013 edition of Nature Communications, has resulted in the creation of more than 60 new families of materials so far, with the potential for many more. The key catalyst in the process is recyclable, making it a 'green' technology.
Four patent applications related to the discovery are pending. VeruTEK, a chemical innovations company based in South Windsor, Conn., has secured rights to some of the materials.
"This is definitely the most exciting project I've been involved in over the past 30 years," says Board of Trustees Distinguished Professor Steven L. Suib, the project's principal investigator. "What we've done is similar to discovering a new insect, only now there is a series of families of these things that can be discovered. That's pretty cool."
The research is the first major work to come out of the University's new GEMS Center of Excellence. The center, which gets its name from the acronym Green Emulsions, Micelles and Surfactants, is located in the Department of Chemistry in the College of Liberal Arts and Sciences.
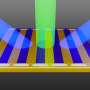
Suib's research involves the creation of uniform, or monomodal, mesoporous metal oxides using transition metals such as manganese, cobalt, and iron. Mesoporous describes the size of the pores in the material. In this case, they are between 2 and 50 nanometers in diameter and are evenly distributed across the material's surface, similar to what one might see if a pin is used to poke numerous holes in a material. Only the UConn process allows scientists to use nitric oxide chemistry to change the diameter of the "pin," in order to change the size of the holes. This unique approach helps contain chemical reactions and provides unprecedented control and flexibility.
"Professor Suib and his colleagues report an unexpected and novel route to generation of mesoporous metal oxides," says Prabir Dutta, distinguished university professor of chemistry and biochemistry at The Ohio State University. "Professor Suib's discovery and the extension of mesoporosity to a much broader range of metal oxides is bound to push this area to new heights, with all sorts of potential applications, making this study a most important development in materials science."

Having materials with uniform microscopic pores allows targeted molecules of a particular size to flow into and out of the material, which is important in such applications as adsorption, sensors, optics, magnetic, and energy products such as the catalysts found in fuel cells.
"When people think about these materials, they think about lock-and-key systems," says Suib. "With certain enzymes, you have to have pores of a certain size and shape. With this process, you can now make a receptacle for specific proteins or enzymes so that they can enter the pores and specifically bind and react. That's the hope, to be able to make a pore that will allow such materials to fit, to be able to make a pore that a scientist needs."
For the past 20 years, scientists have relied on a long-standing, water-based procedure for making mesoporous materials that was first developed by Mobil Oil. That procedure, although groundbreaking when it was discovered, has limitations. The size of the pores in the material is difficult to manipulate; the walls of the resulting mesoporous structures are amorphous; and the stability of the underlying system weakens when exposed to high heat, limiting its use. The process also only works best when using silicon or titanium, as opposed to other metals of the periodic table.
UConn's chemists took another route, choosing to replace the water-based process with a synthetic chemical surfactant similar to a detergent to create the mesopores. By reducing the use of water, adding the surfactant, then subjecting the resulting nanoparticles to heat, the research team found that it could generate thermally-controlled, thermally-stable, uniform mesoporous materials with very strong crystalline walls. The mesopores, Suib says, are created by the gaps that are formed between the organized nanoparticles when they cluster together. The team found that the size of those gaps or pores could be tailored – increased or decreased – by adjusting the nanostructure's exposure to heat, a major advancement in the synthesis process.
"Such control of pore-size distribution, enhanced pore volumes, and thermal stabilities is unprecedented …," the team wrote in its report.
Perhaps just as importantly, the team found that the process could be successfully applied to a wide variety of elements of the periodic table. Also, the surfactant used in the synthesis is recyclable and can be reused after it is extracted with no harm to the final product.
Aware of the significance of its findings, Suib's team deliberately waited to publish its report until it had verified different applications using a variety of periodic elements. Even now, the team believes it has only scratched the surface.
"We developed more than 60 families of materials," says Suib. "For every single material we made, you can make dozens of others like it. You can dope them by adding small amounts of impurities. You can alter their properties. You can make sulfides in addition to oxides. There is a lot more research that needs to be done."
The UConn research was funded by the U.S. Department of Energy's Basic Energy Sciences division through a $420,000 grant over three years. Suib says one particular member of the research team, Altug S. Poyraz, an 'enormously talented' graduate student pursuing his doctoral degree at UConn, was instrumental to the project's success. Poyraz spent countless weeks patiently exploring different approaches to the process until the team found success.
"He's truly a unique grad student and probably the best synthetic chemist I've ever seen," said Suib, who also serves as director of UConn's Institute of Materials Science.
Suib believes the process will be attractive to industry because it is simple, cost-effective, and green.
Journal information: Nature Communications
Provided by University of Connecticut