Amoeboid swimming - crawling in a fluid
Researchers from CNRS, Inserm, and Université Joseph Fourier - Grenoble have developed a particularly simple model that reproduces the swimming mechanism of amoebas. They show that, by changing shape, these single cell organisms propel themselves forward in a viscous fluid at the same speed as when they crawl on a solid substrate. This work has recently been published in the journal Physical Review Letters.
The way microorganisms swim is fundamentally different to that of fish since, at their scale, viscosity effects dominate and make fins totally inefficient. Various strategies are employed. The majority of such organisms propel themselves forward by beating their flagella or cilia while others, such as amoebas, deform their bodies in the same way as they would for crawling. However the efficiency of this method of propulsion remains poorly understood.
Physicists from the Laboratoire Interdisciplinaire de Physique (LIPhy, CNRS/Université Joseph Fourier Grenoble), Oslo University and the Institut Albert Bonniot (Inserm/Université Joseph Fourier – Grenoble) have elucidated the key elements of this method of locomotion by analyzing a simplified theoretical model.
They determined the necessary morphological deformations and the speed of propulsion and showed that incompressibility of the cell membrane is essential.
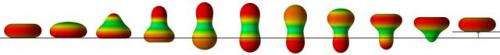
To conduct this study, the researchers modeled the cell using an inextensible fluid membrane (in other words, able to deform while maintaining its membrane area) containing a viscous fluid and located within a viscous fluid. Surface deformations in this model are uniquely due to forces perpendicular to the surface of the membrane. Among all the possible deformations, the physicists favored those that maintain symmetry of revolution around the axis of movement. Cell deformations induce stresses in the external fluid, which, in return, exerts a force on the cell. To simulate a swimming motion, the researchers considered elementary movements during which the forces exerted on the surface remain constant. These forces alter the shape of the cell and calculations show that motion depends solely on these shapes and not on the speed of movement.
Swimming is thus uniquely determined by the succession of shapes adopted by the cell and the distance covered only depends on the geometry of the surfaces. The model described reproduces certain swimming cycles observed in nature. It will certainly improve our knowledge of cell mobility and make it possible to envisage novel types of artificial micro-swimmers.
More information: Amoeboid Swimming: A Generic Self-Propulsion of Cells in Fluids by Means of Membrane Deformations, A. Farutin, S. Rafaï, D. K. Dysthe, A. Duperray, P. Peyla and C. Misbah. Published on the 27 November 2013 in Physical Review Letters.
Journal information: Physical Review Letters
Provided by CNRS