Molecular mirror images assigned: Safer drugs thanks to a new solution to a 150-year-old chemistry problem?

Just like gloves, molecules come in so-called left-handed and right-handed versions. Until now, however, it could be determined only with great difficulty whether a certain molecule is right-handed or left-handed. In medicine, this would be a big step forward because, for example, the unwanted side effects of drugs could be avoided. In the current issue of the journal Science, a team of researchers from Germany, Canada and Switzerland reports a new solution to a 150-year old problem.
The phenomenon of molecules with handedness is well known in yogurt: the bacterial cultures produce levo- or dextrorotatory lactic acid; of these two types, one form has a more favorable influence on intestinal flora than the other. In other substances, the second version is less harmless and causes damage: for example, while one form of penicillamine is effective against arthritis, its mirror image is more toxic.
"Enlargement" due to explosion
But how can we determine whether molecules are right- or left-handed? Until now, it was possible to directly determine handedness only in solid, crystalline substances using a special process in which the crystal structure was analyzed using X-rays. "The problem with this method is that it is not easy to crystallize every substance or to simply incorporate it into a suitable crystal. Therefore, we investigated a method in which the handedness can be determined directly in the gas phase" explains Prof. Robert Berger of the Clemens-Schöpf Institute at TU Darmstadt.
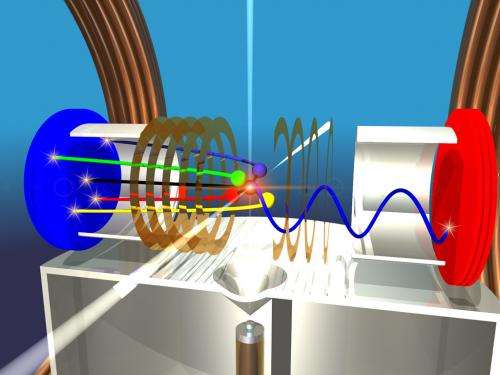
It is based on "enlargement" due to explosion. Imagine inflating a rubber glove until it bursts. If you follow the trajectory of each finger in the opposite direction, you obtain the original glove and can see whether it is a right or left glove. The researchers proceeded in a similar way for the molecules that they studied.
Their test subject was bromochlorofluoromethane, an easily evaporable liquid carbon compound with four different bonding partners. The molecule has the shape of a tetrahedron, with carbon in the middle and hydrogen, bromine, chlorine and fluorine at the corners. The handedness results from the distribution of the bonding partners on the four corners.
To determine this, an intense laser beam is used to remove one type of electron from all the atoms at once. The molecule, which now has five positive charges, explodes due to the high repulsion between the positively charged modules. The particles then collide with a detector that determines the duration of the flight and the location of the impact, thus allowing the flight path to be established. From this, the spatial arrangement of the atoms in the molecule before the explosion can be reconstructed.
A variety of applications
"This method opens new perspectives for the investigation and analysis of handed molecules in physics, chemistry and pharmaceutics" predicts Dr. Markus Schöffler of the Institute of Nuclear Physics at Goethe University. For example, drugs could be produced in which only the molecules of the desired handedness are present. As a result, the dosage could be reduced. But other industries would also benefit: in carvone, a component of essential oils, for example, the handedness determines whether it smells like spearmint or caraway. In other substances, the flavor changes from bitter to sweet, for instance.
"However, being able to work only with the desired molecular type is a long-term goal" Berger points out. "We now have for the time being proved, for the textbook example of a handed substance, that direct assignment in the gas phase is possible."
More information: Pitzer, M. et al. Direct Determination of Absolute Molecular Stereochemistry in Gas Phase by Coulomb Explosion Imaging, Science, 2013. DOI: 10.1126/science.1240362
Journal information: Science
Provided by Technische Universitat Darmstadt