Using a form of 'ice that burns' to make potable water from oil and gas production
In the midst of an intensifying global water crisis, scientists are reporting development of a more economical way to use one form of the "ice that burns" to turn very salty wastewater from fracking and other oil and gas production methods into water for drinking and irrigation. The study on the method, which removes more than 90 percent of the salt, appears in the journal ACS Sustainable Chemistry & Engineering.
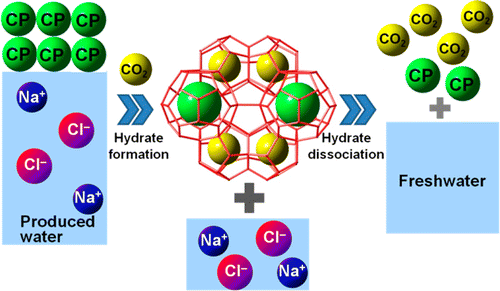
Yongkoo Seol and Jong-Ho Cha explain that salty wastewater is a byproduct of oil and gas production, including hydraulic fracturing, or fracking. These methods use water and produce as a byproduct almost 10 barrels of salty water for every barrel of oil. That water could help people in water-stressed regions. But it can't be desalinated economically with traditional methods. Seol and Cha knew that an alternative called "gas hydrate desalination" showed promise. A gas hydrate consists of only water and a gas such as methane, the stuff of natural gas. Thus, when hydrates form, salts and other impurities are left behind. When the hydrate breaks down, the gas and pure water are released. However, forming the gas hydrate used in desalination required costly chilling of the water to 28 degrees Fahrenheit. Seol and Cha sought to develop a less costly version of the method, which involves a variation on methane hydrates, chunks of ice retrieved from deep below the sea that burst into flame when brought to the surface.
They describe development and laboratory testing of a new type of gas hydrate desalination technique. They formed the hydrates from water and carbon dioxide with the gases cyclopentane and cyclohexane, which made the method work more efficiently. It removed more than 90 percent of the salt compared to 70 percent with the previous gas hydrate technique. And the process works at near-room temperature, reducing the need for chilling.
More information: "Increasing Gas Hydrate Formation Temperature for Desalination of High Salinity Produced Water with Secondary Guests" ACS Sustainable Chem. Eng., Article ASAP. DOI: 10.1021/sc400160u
Abstract
We suggest a new gas hydrate-based desalination process using water-immiscible hydrate formers; cyclopentane (CP) and cyclohexane (CH) as secondary hydrate guests to alleviate temperature requirements for hydrate formation. The hydrate formation reactions were carried out in an isobaric condition of 3.1 MPa to find the upper temperature limit of CO2 hydrate formation. Simulated produced water (8.95 wt % salinity) mixed with the hydrate formers shows an increased upper temperature limit from ?2 °C for simple CO2 hydrate to 16 and 7 °C for double (CO2 + CP) and (CO2 + CH) hydrates, respectively. The resulting conversion rate to double hydrate turned out to be similar to that with simple CO2 hydrate at the upper temperature limit. Hydrate formation rates (Rf) for the double hydrates with CP and CH are shown to be 22 and 16 times higher, respectively, than that of the simple CO2 hydrate at the upper temperature limit. Such mild hydrate formation temperature and fast formation kinetics indicate increased energy efficiency of the double hydrate system for the desalination process. Dissociated water from the hydrates shows greater than 90% salt removal efficiency for the hydrates with the secondary guests, which is also improved from about 70% salt removal efficiency for the simple hydrates.
Provided by American Chemical Society


















