Nanoparticles for controlled drug release
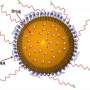
Scientists from CIC bioGUNE and the Laboratoire de Chimie des Polymères Organiques (LCPO) in Bordeaux have jointly undertaken a project to develop "smart" nanoparticles. These polymeric particles act as "nanomissiles" against pre-determined targets and allow the drug to be released in a spatially and temporally controlled manner, only releasing their "load" where it is needed. Release of the medication is controlled by applying a localised magnetic field.
Chemists from the LCPO were responsible for generating the nanoparticles, which are approximately the same size as a virus, whereas researchers from CIC bioGUNE were responsible for assessing their efficacy in a cell culture model. This study has been published this week in the Journal of Controlled Release.
The technique developed increases treatment efficacy as it allows the medication to be deposited directly in the affected organ, thereby minimising side effects in other organs. The side effects of any chemotherapy treatment generally arise due to the toxic effects of the drugs administered on healthy tissue (for example hair loss). Indeed, these side effects often mean that the optimal dose cannot be used as it would be far too toxic for the patient.
The system developed by the LCPO/CIC bioGUNE team should allow the drug to be released inside the organ when required. The nanoparticles used to transport the medication are iron oxide-containing polymers. As such, a series of "pores" on the surface of the polymer open up when a magnetic field is applied, thus releasing the drug.
This localised release of the medication should reduce its effect on healthy tissue and may mean that the dose received by cancer cells can be increased. The benefits of this method are therefore decreased side effects and increased treatment efficacy. In the words of the CIC bioGUNE researcher Edurne Berra, "application of a localised magnetic field allows release of the drug and increases its cytotoxic effects on cancer cells".
This work has used doxorubicin, a drug widely used in anti-cancer chemotherapy, as a model. However, its conclusions are likely to be simply the starting point for the development of new, smart release systems for other drugs.
Furthermore, as also noted by Dr. Berra, "the system studied should allow drugs other than doxorubicin to be encapsulated, and it may even prove possible to add molecules that recognise a specific type of cancer cell. It may also find a use for the magnetic resonance-based diagnosis of cancer and even for theragnosis, in other words simultaneous diagnosis and therapy".
More information: Oliveira, H. et al. Magnetic field triggered drug release from polymersomes for cancer therapeutics, Journal of Controlled Release. Volume 169, Issue 3, 10 August 2013, Pages 165–170. www.sciencedirect.com/science/ … ii/S0168365913000308
Journal information: Journal of Controlled Release
Provided by Centro de Investigacion Cooperativa en Biociencias