Controlled and targeted release of drugs

(Phys.org)—Researchers have discovered a method that allows for the controlled release of an active agent on the basis of a magnetic nanovehicle. The research, conducted by EPFL, the Adolphe Merkle Institute and the University Hospital of Geneva, opens up new possibilities for the develop-ment of target.
Certain drugs are toxic by nature. For example, anti-cancer drugs developed to kill diseased cells also harm healthy ones. To limit the side effects of chemotherapy, it would be a great step forward if it were possible to release a drug only in the affected area of the body. In the context of the National Research Programme "Smart Materials" (NRP 62) - a cooperation between the SNSF and the Commission for Technology and Innovation (CTI) - researchers of ETH Lausanne, the Adolphe Merkle Institute and the University Hospital of Geneva have discovered a method that might represent an important step towards the development of an intelligent drug of this kind. By combining their expert knowledge in the areas of material sciences, biological nanomaterials and medicine, they were able to prove the feasibility of using a nanovehicle to transport drugs and release them in a con-trolled manner.
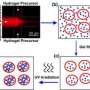
This nanocontainer is a liposome, which takes the shape of a vesicle. It has a diameter of 100 to 200 nanometers and is 100 times smaller than a human cell. The membrane of the vesicle is composed of phospholipids and the inside of the vesicle offers room for the drug. On the surface of the liposome, specific molecules help to target malignant cells and to hide the nanocontainer from the immune system, which might otherwise consider it a foreign entity and seek to destroy it. Now the researchers only needed to discover a mechanism to open up the membrane at will.
Nano effect
This is exactly what the researchers succeeded in doing. How they did it? By integrating into the liposome membrane superparamagnetic iron oxide nanoparticles (SPION), which only become magnetic in the presence of an external magnetic field. Once they are in the field, the SPION heat up. The heat makes the membrane permeable and the drug is released. Researchers proved the feasibility of such a nano-vehicle by releasing in a controlled manner a coloured substance contained in the liposomes. "We can really talk of nanomedicine in this context because, by exploiting superparamagnetism, we are ex-ploiting a quantum effect which only exists at the level of nanoparticles," explains Heinrich Hofmann of the Powder Technology Laboratory of EPFL. SPION are also an excellent contrast agent in magnetic resonance imaging (MRI). A simple MRI shows the location of the SPION and allows for the release of the drug once it has reached the targeted spot.
Designed for medical practice
"To maximise the chances of discovering an effective treatment, we focused on nanocontainers, which would be readily accepted by doctors," adds Heinrich Hofmann. This strategy limits the range of possibilities. Liposomes, which are already used in a number of drugs on the market, are composed of natural phospholipids which can also be found in the membranes of human cells. To open them up, researchers focused on SPION, which had already been the subject of numer-ous toxicological studies. More efficient materials were ignored because little or nothing was known about their effects on humans. In terms of shape, another important parameter of magnetism, they chose to use only spherical nanoparticles, which are considered safer than fibrous shapes. The intensity and frequency of the magnetic field needed to release the active agent are compatible with human physiology.
The combination of these parameters presented the researchers with another challenge: to reach a temperature sufficiently high to open up the liposomes, they were forced to increase the size of the SPION from 6 to 15 nanometres. The membrane of the vesicles has a thickness of only 4-5 nanometres. Then the masterstroke: the research group of Alke Fink at the Adolphe Merkle Institute was able to regroup the SPION in one part of the membrane. This also made MRI detection easier. Before starting in-vivo tests, the researchers aim to study the integration of SPION into the liposome membrane in greater detail.
Provided by Ecole Polytechnique Federale de Lausanne