Team observes real-time charging of a lithium-air battery

One of the most promising new kinds of battery to power electric cars is called a lithium-air battery, which could store up to four times as much energy per pound as today's best lithium-ion batteries. But progress has been slow: The nature of the electrochemical reactions as these batteries are charged remains poorly understood.
Researchers at MIT and Sandia National Laboratories have used transmission electron microscope (TEM) imaging to observe, at a molecular level, what goes on during a reaction called oxygen evolution as lithium-air batteries charge; this reaction is thought to be a bottleneck limiting further improvements to these batteries. The TEM technique could help in finding ways to make such batteries practical in the near future.
The work is described in a Nano Letters paper by Robert Mitchell, who recently received a PhD in materials science and engineering from MIT; mechanical engineering PhD student Betar Gallant; Carl Thompson, the Stavros Salapatas Professor of Materials Science and Engineering; Yang Shao-Horn, the Gail E. Kendall Associate Professor of Mechanical Engineering and Materials Science and Engineering; and four other authors.
Oxidation in action
The new observations show, for the first time, the oxidation of lithium peroxide, the material formed during discharge in a lithium-air battery. At high charging rates, this oxidation occurs mostly at the boundary between the lithium peroxide and the carbon substrate on which it grows during discharge—in this case, multiwall carbon nanotubes used in the battery electrode.
The confinement to this interface, Shao-Horn says, shows that it is the resistance of lithium peroxide to a flow of electrons that limits the charging of such batteries under practical charging conditions.
An electrolyte-coated probe tip serves as the opposing electrode for removing lithium ions during charging, as electrons flow through the nanotube framework to the external circuit. During charging, the lithium peroxide particles shrink beginning at the nanotube-peroxide interface, showing that oxidation occurs where it is easiest to remove electrons.
"The lithium transport can keep up," Shao-Horn says, which indicates that electron transport could be a critical limit on charging of batteries for electric vehicles.
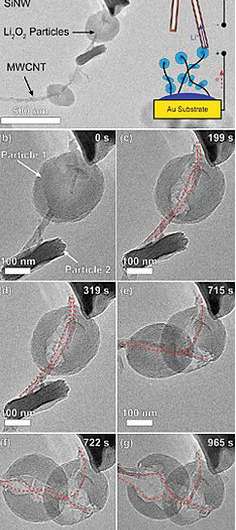
Faster charging
In fact, the rate of lithium peroxide oxidation in these experiments was approximately 100 times faster than the charging time for laboratory-scale lithium-air batteries, and approaches what is needed for applications. This demonstrates that if these batteries' electron-transfer characteristics can be improved, it could allow for much faster charging while minimizing energy losses.
"This provides insights into how to design the air electrode," Shao-Horn says. "To our knowledge, this is the first direct evidence that electron transport is limiting the charging."
Gallant says this finding suggests that lithium-air battery performance would improve if electrodes had a high-surface-area structure to maximize contact between lithium peroxide and the carbon required to transport electrons away during charging.
The "very critical next step," Shao-Horn says, will be to measure actual currents during charging. Her team is working with researchers at Sandia National Laboratories, some of whom were co-authors of this paper.
Jie Xiao, a researcher at Pacific Northwest National Laboratory who was not involved in this research, says, "This work has identified the key limiting condition, electron transport … providing a critical contribution."
Xiao adds, "This is a great example of how fundamental research can significantly improve our understanding to resolve challenges in practical devices. The information provided in this paper will benefit the rational design of the air electrode of lithium-air batteries. … This research is of high quality and will attract broad interest."
More information: "In Situ Transmission Electron Microscopy Observations of Electrochemical Oxidation of Li2O2" pubs.acs.org/doi/abs/10.1021/nl400731w
Journal information: Nano Letters
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.