Bacteria with vuvuzelas: Microbes use a channel protein as a syringe for toxins
The bacterium Photorhabdus luminescens is a constant companion of some roundworms. These worms assault insect larvae, thereby infecting them with the bacteria; the pathogens then attack the cells of their victims with a deadly cocktail of various toxins. Scientists at the Max Planck Institute of Molecular Physiology in Dortmund working together with colleagues from Freiburg University and Jacobs University Bremen, have discovered that the bacteria use an important toxin complex like a syringe. It makes its way into the host cells via constricted vesicles in the cell membranes, and modifies their structure from within. Part of the toxin complex then forces its way inside the cell through the vesicle membrane by means of a vuvuzela-like protein channel, and kills the cell.
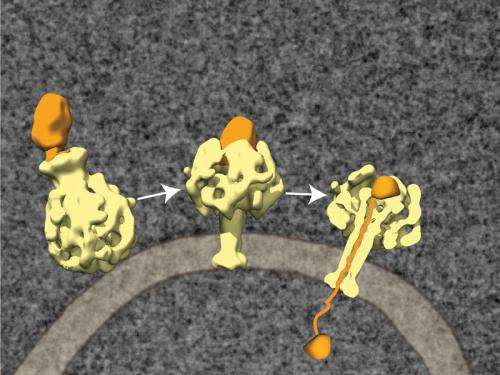
Important toxins of Photorhabdus luminescens are counted among the ABC toxins, which consist of the three protein components TcA, TcB and TcC. The toxin complex first docks at receptor molecules on the membrane of the host cell and is sucked inside the cell in small membrane blisters called vesicles. The TcC components then make their way into the cell fluid and demolish the cell's protein skeleton. What has remained unclear to date, however, was how the protein managed to get through the vesicle membrane.
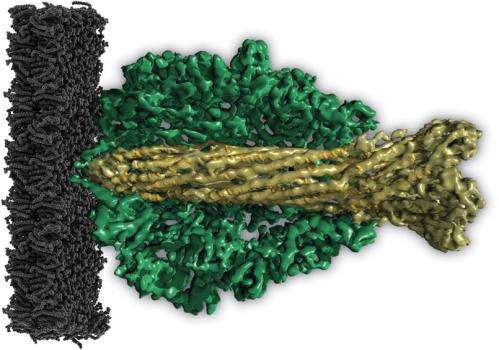
Now, for the first time, scientists have been able to decode the structure of Photorhabdus luminescens' ABC toxins using cryoelectron microscopy and single particle analysis. This shows that the bacterium's TcA protein consists of five subunits that together form the shape of a bell. "Inside the bell, the subunits form a channel that has one wide and one narrow aperture, so that it looks like the notorious vuvuzela horn used by South African football fans", explains Stefan Raunser of the Max Planck Institute of Molecular Physiology.
As soon as the pH value of the environment rises or falls, for example when the fluid in the vesicles turns acid, the outer shell of the toxin opens, unblocking the central channel. "Now the channel is pushed through the cell membrane like the needle of a syringe", says Raunser. TcB and TcC are drawn into the area between the channel and the shell, where TcC is unpacked and loses its original structure. "It may be that a drop in electrical tension or some specific unpacking proteins such as TcB are necessary for TcC to exit the vesicle and enter the cell, where it can unleash its deadly effects."
The results show that the TcA of roundworm bacteria is similar in shape to the toxins of the plague pathogen and other bacteria. "This may mean that these findings will also reveal the workings of bacteria that induce disease in humans", says Raunser. The findings could also help in the development of pest-resistant crops.
In addition to the ABC toxins, plague pathogens have developed another transport system that also occurs in the pathogens of dysentery and typhus. Known as the type III secretion system, it also looks like a syringe. However, the body of the syringe is embedded in the bacterial membrane, with the needle pointing outwards. The bacteria use these nanosyringes to inject material directly into their host cells.
More information: Gatsogiannis, C. et al. Photorhabdus luminescens toxins use a novel syringe-like injection mechanism for cell entry, Nature, March 20, 2013. DOI: 10.1038/nature11987
Journal information: Nature
Provided by Max Planck Society