Bioscientists develop a biosensor for the early diagnosis of sleeping sickness
Infectious African sleeping sickness is widespread south of the Sahara Desert. Although the around sixty million people residing in tropical Africa run the risk of becoming infected every day, only around four million of them are monitored for the disease by disease-control authorities. TU Darmstadt bioscientists have recently developed a biosensor that provides simple, cost-effective, means for diagnosing incidences of the disease before it breaks out.
Tsetse flies, which remain active around the clock, transmit a single-celled parasite belonging to the species trypanosoma. Those who become infected with such parasites will die if they remain untreated. Treatment at an advanced stage, i.e., weeks to months following infection with the disease, is difficult, since, by then, the parasites will have penetrated the central nervous system, where they cause irreversible harm.
From that stage onward, only highly toxic medications, such as arsenic compounds, are available for treatment. Due to their severe side effects, treatment with such medications must take place in hospitals, and even that might not save patients' lives. Cows can also become infected with the disease. The emaciated domestic animals frequently seen in television reports are usually suffering from it. This devious disease thus also adversely affects the livelihoods of those residing in enhazarded regions.
An old method employing new means
Professor H. Ulrich Göringer, of the Working Group on Molecular Genetics at the TU Darmstadt's Biology Dept., stated that, "We can now determine, from a few drops of blood, whether someone has been infected with the disease before it breaks out." He and his group have developed a new type of synthetic biosensor for its diagnosis. They are the first to combine potentiometry, a well-established method for detecting substances by measuring changes in electrostatic potentials, with biological molecules that serve as diagnostic probes.
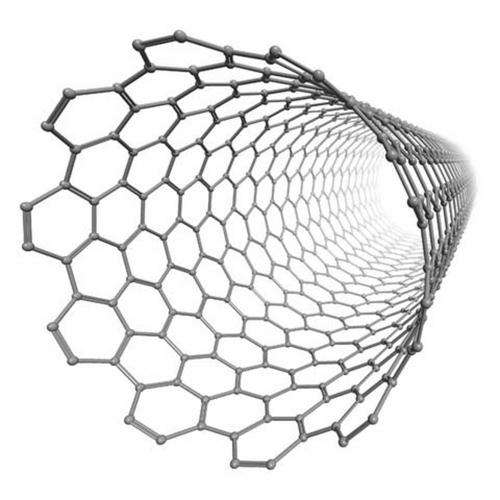
The molecules involved are ionic, i.e., carry an electric charge that can change when they react with other molecules. Those changes are detected by a microelectrode immersed in a sample solution, e.g., blood. The microelectrode is configured from carbon nanotubes, to which the biological molecules involved are coupled. Their bonding to the parasites causing the disease is an indicator of the presence of the latter.
The biological molecules involved are aptamers that are chemically produced and comparable to the DNA molecules and RNA molecules that transmit genetic information. The TU Darmstadt group has developed an aptamer that, due to its molecular structure, bonds to the pathogen, the trypanosomes that cause sleeping sickness, with a high degree of specificity.
If a microelectrode configured from carbon nanotubes and the aptamer is wetted with a test solution containing such trypanosomes, the aptamers will bond to the trypanosomes, which will cause a change in the charges carried by the molecules, and thus a change in the microelectrode's electrostatic potential. As Prof. Göringer put it, "Since the nanotubes are highly electrically conductive, our detection method is ultrasensitive. Presence of just a few trypanosomes in a single drop of blood will suffice for their detection."
Circumventing the parasites' tricks
However, trypanosomes have a high capacity for undergoing transformations. In order to avoid attacks by the human immune system, which can form antibodies that combat intruders, they enshroud their cell membranes in a layer of defender molecules. The latter molecules, which are termed "variable surface antigens" ("variable surface glycoproteins" (VSG)), are intended to deceive the immune system's antibodies.
Before the immune system has had a chance to adjust to a particular VSG, the trypanosomes switch to another VSG in order that the antibodies will no longer be able to recognize them. Around 1,000 types of trypanosomes are capable of causing sleeping sickness, which is enough to allow them to multiply, unimpeded by attacks by the immune system.
The TU Darmstadt group has, nevertheless, found a means for turning the tables and deceiving the trypanosomes involved. Changes in their VSG are confined to a variable "tail zone" on their VSG, and do not affect their overall structures. However, the RNA aptamers adhering to the carbon nanotubes bond to the trypanosomes' tails, which remain unaffected by changes in their VSG. Although their tails are enclosed within their VSG shrouds, the aptamers are small enough to sneak into their VSG shrouds.
Chips that will allow simultaneously diagnosing several diseases
Göringer is very enthusiastic about his group's discoveries: "The metrological method involved is very simple. No biochemical preprocessing is necessary, and just a few drops of blood are enough to allow detecting the parasites involved." Although there are very many other ionic, that is, electrically charged, molecules present in blood, thanks to a collaboration with the Harvard Medical School in Boston, the TU Darmstadt group has succeeded in reducing the interferences generated by such molecules to "a sort of noise."
The carbon-nanotube/aptamer composites involved may be sprayed onto paper or plastics utilizing standard processes, where the manufacture of diagnostic kits would currently cost around five U.S. Dollars application. Göringer forecasted that, "We hope to have enabled their mass production within two to three years, which will make the method less expensive." Cost reductions would also allow diagnosing domestic animals for sleeping sickness and bolster the livelihoods of the people involved.
Cutting their manufacturing costs has spawned another idea that Göringer summarized as follows: "We can envision producing biosensor microelectrodes similar to those employed in the method developed at the TU Darmstadt in the form of 'printed electronics,' simple metrological chips or test strips, fabricated employing standard printing methods." That would allow cutting the cost per diagnosis to one to two U.S. Dollars, which would bring it into accord with the cost envisioned by the World Health Organization (WHO) for the diagnosis of "neglected diseases."
As might be expected, there is not much interest in this particular neglected disease among members of the medical-diagnostics industry. Nevertheless, in spite of all the negative factors, the TU Darmstadt group believes that it will succeed in finding industrial partners interested in mass-producing products resulting from their researches, particularly since their diagnostic method can be extended to cover any other disease. As Göringer put it, "As a next step, we intend to develop a chip that will allow simultaneous diagnoses of several infectious diseases. In general, our method may be applied to the diagnosis of any disease. The sole prerequisite is an ability to generate aptamers that bond exclusively to certain pathogen molecules, and that is where our expertise comes in."
Provided by Technische Universitat Darmstadt















