November 13, 2012 feature
The road also taken: Alternative and hidden pathways in biological electron transfer
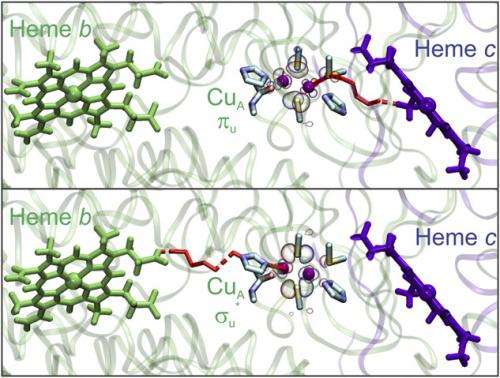
(Phys.org)—Photosynthesis, cellular respiration and many other biological processes have something in common: long-range electron transfer (ET), the simplest known chemical reaction that is known to occur efficiently high over distances up to 20 Å. However, determining the ways in which biological electron transfer is optimized has been a subject of considerable discussion. Recently, scientists at Universidad Nacional de Rosario and Universidad de Buenos Aires, Argentina conducted a spectroscopic, electrochemical, and theoretical study that suggests a unique role for alternative or "invisible" electronic ground states corresponding to directional electron transfer. Specifically, they describe a biological redox metal center (CuA) in which thermal fluctuations in Thermus thermophilus may populate two alternative ground-state electronic wave functions optimized for electron entry and exit, respectively, through two different and nearly perpendicular pathways.
Professors Daniel H. Murgida and Alejandro J. Vila outline the challenges they and their fellow researchers, including Luciano A. Abriata and Damián Álvarez-Paggi, encountered when conducting their study. "Perhaps the main challenge associated with our discovery was to surmount our own preconceived hypothesis of how the ET reactions of CuA-containing enzymes should occur," Murgida tells Phys.org. "At the beginning there was no place in our minds for the picture of alternative electronic states operating for electron entry and exit. We had a large collection of experimental and theoretical results, but they just didn't add up – evidence seemed contradictory or incomplete at best. In fact, some pieces of the puzzle that tied up everything quite nicely appeared later on in our investigation."
Their seed ideas, Vila continues, were triggered by paramagnetic NMR spectroscopy and quantum mechanical calculations. Once they identified and characterized these two alternative ground states, and realized that thermal fluctuations and small external perturbations determined their relative populations, they began to comprehend much of their spectroscopic and electrochemical data.
"Suddenly," Murgida says, "everything fit. Then we began wondering, were we really the only ones who have seen this? We went back through the literature, and the answer was no: Many researchers before us obtained pieces of what we now know as evidence in favor of the existence of alternative ground-states in CuA centers – but just as it happened to us at first, they simply didn't realize what the data meant." After their findings, Murgida adds, the data can be reinterpreted in terms of the many different perturbations that may switch the population of either state, thereby reinforcing the notion of this mechanism being biologically relevant.
In addressing these challenges, the researchers utilized a range of techniques. "We did lots of spectroscopy, such as NMR, UV/Vis, EXAFS and Resonance Raman," Murgida explains. "Then we probed the redox functionality using protein film voltammetry – and, as mentioned, we also performed many computational calculations at the levels of classical and quantum mechanics. "This allows for high-resolution definition of activity across a wide and continuous potential range, complementing the information available from more traditional methods of analysis with resolution in the time and concentration domains," Murgida details. "In addition, PFV affords high sample economy, films typically contain less than or equal to 1 picomole of protein, and simply transferring a film between solutions of different composition allows changes of reactivity, and their reversibility, to be readily established."
(Protein film voltammetry (PFV) is an electrochemical technique that provides thermodynamic and kinetic descriptions of electron transfer and coupled chemistry occurring within redox proteins, and moreover has the entire electroactive sample under precise potential control at all times in an experiment.)
"Of great importance is that all of the experiments and simulations were performed both for the wild type protein and for single point mutants that were specifically designed and produced in our labs," Vila continues. "These mutations allowed us to perturb the electronic structure of the protein active site while preserving its native fold. Understanding exactly how the electronic structure was altered, and identifying the thermodynamic and kinetic ET properties that changed, were the key insights in our study."
Murgida also discussed key results suggested by the study's findings – specifically, the role of alternative or invisible electronic ground states in directional electron transfer, as well as alternative ways through which protein–protein interactions and membrane potential may optimize and regulate electron–proton energy transduction. "At room temperature, every molecule exists in a form of minimal electronic energy that is usually called the fundamental or ground state. Promotion to quantum mechanically-allowed states of higher energy – that is, excited states – usually requires a large amount of energy that is not thermally available under these conditions." As a result, except for light-activated processes such as photosynthesis, chemical reactions usually involve the ground state only.
"In our work, we found that, for the CuA site, very small structural fluctuations and small perturbations change the nature of the electronic ground state, and that the new ground state has very different quantum properties and, thereby, reactivity," Vila continues. "Unlike the case of electronically-excited states, the energy gap between these two ground states is small, meaning that both are thermally accessible. Moreover, it can be finely tuned either by small structural perturbations or by the presence of electric fields such as those generated by the membrane potential, thus altering the relative populations of states."
Interestingly, while one of the states is optimized for electron entry from Cytochrome c (Cyt) – a natural electron carrier in respiration – the other is optimized for electron exit, towards molecular oxygen (O2), the final electron acceptor in respiration). In its idle state, both researchers point out, the CuA site is mainly in the state not optimized for electron entry, which we therefore call invisible. However, they add, subtle perturbations that take place when forming the inter-protein Cyt- CuA complex shift the balance towards the alternative invisible state, thus facilitating electron transfer from Cyt to CuA, and subsequent transfer to the next redox center. In other words, the CuA site behaves as an electron hub, directing efficient electron flow by using these two different electronic pathways.
Murgida notes that their findings have implications for areas of research beyond their own. "Both basic and applied research may find these results useful. Our research shows that there are still many aspects of biological electron transfer that haven't been unveiled yet, and may push the study of electron/proton energy transduction further. On the other hand, our findings may exert a profound impact in the development of protein-based electronic devices such as biosensors, suggesting new ways in which electron transfer reactions may be finely tuned in order to design more efficient devices."
Not surprisingly, then, the researchers are looking at ways to extend their work. "For us, there's not only one next step," Vila concludes. "Forthcoming challenges involve three vital questions: Can we induce this ground state switch with even more subtle perturbations farther away from the metal site? Can we engineer a CuA site in which the naturally invisible state becomes more stable than the visible one? Can we elicit this two-state switching by protein-protein interactions? There are many things we'd like to try right now."
More information: Alternative ground states enable pathway switching in biological electron transfer, PNAS ,October, 23, 2012 vol. 109 no. 43, 17348-17353, doi:10.1073/pnas.1204251109
Journal information: Proceedings of the National Academy of Sciences
Copyright 2012 Phys.org
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.