Field-proven meter rapidly determines carbon dioxide levels in groundwater
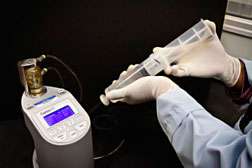
NETL and West Virginia University collaborators discovered that a standard beverage industry carbonation meter used with a modified field protocol accurately determined the amount of CO2 dissolved in natural springs and mine waters within the range of 0.2 - 35 millimole (a mole is a measurement for chemicals, thus a millimole is one thousandth of a mole) of CO2.
The meter, which measures dissolved CO2 based on temperature and pressure changes determined during sample volume expansion, offers a new way to measure dissolved CO2 rapidly and reproducibly in a wide range of natural waters, which is critical when investigating possible leakage from carbon sequestration sites. Estimates of dissolved CO2 concentrations using conventional titration approaches are time-consuming, and results can vary widely because of various sources of error (e.g., rapid degassing, low alkalinity, non-carbonate alkalinity).
Carbonation meter measurements compared well with those obtained with more conventional approaches, but were immediate and avoided errors introduced by alkalinity. The portable meter is practical for use in difficult terrain, and the technique proved useful for studying aquatic systems in which CO2 degassing drives geochemical changes that result in mineral precipitation and deposition.
More information: Selected results of this research appear in Elsevier’s Journal of Hydrology (doi:10.1016/j.jhydrol.2012.03.015).
Provided by West Virginia University