Neutrons explain how haemoglobin evolution in red blood cells helped the duck-billed platypus respire
Scientists have explained the evolutionary history of haemoglobin using what might seem an unlikely array of samples.
Researchers focused the world’s most intense neutrons beams on the oxygen-carrying protein from a human, a duck-billed platypus, a chicken and a salt-water crocodile to explain how it has adapted to different body temperatures within different species. The results of research at the Institut Laue-Langevin (ILL), Aachen University of Applied Sciences (FH Aachen), the Australian Nuclear Science and Technology Organisation, the Centre National de la Recherche Scientifique (CNRS) in Paris and the FRMII facility in Germany could lead to interesting developments in bio-engineering and biomedical research.
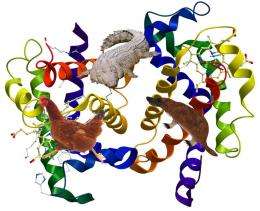
Haemoglobin is an iron-rich protein found in all vertebrates that carries oxygen from the lungs to the rest of the body. The stiffness of human haemoglobin is ideally suited to our own body temperature– 37 degrees Celsius. Research by Gerhard Artmann at FH Aachen showed that at this temperature it partially unfolds, softening sufficiently for oxygen molecules to penetrate its four iron atoms without completely compromising its structural integrity and collapsing.
Scientists have recently looked at whether this loss of structural integrity occurred in all vertebrate haemoglobin and how it varied with the different body temperatures of different species. They looked at a number of species including a platypus, which has the lowest body temperature of the vertebrates (34.0C) and a bird, the common chicken, which has a very high body temp (42.80C). These studies confirmed that each species’ haemoglobin is designed to unfold at exactly its own body temperature suggesting an evolutionary adaptation. However the structural make-up of the different haemoglobins studied was identical. The question therefore remained, where was the temperature-sensing part of the haemoglobin that was driving this evolutionary adaptation?
The key to this mystery, identified by a new paper published in the Journal of the Royal Society Interface, are chemical differences between the different haemoglobins, which lead to similar molecular structures but different dynamics, i.e. different stiffness and flexibility. Whilst all are composed of amino acids, the types of amino acid and the order in which they are found varies.
The flexibility and stiffness of the different haemoglobins were measured by neutron scattering experiments at the ILL and the FRMII facility in Garching. Neutron scattering was chosen as it can accurately measure the movement within the complex structures without destroying the samples, which were very delicate and sensitive to radiation. To find out which amino acids were the source of the stiffness variation, the group of researchers performed complimentary computer simulations at the CNRS in Paris.
The search focused on amino acids on either side of small holes in the haemoglobin, known as cavities, which were known to play a role in regulating the absorption of oxygen. This latest research shows how the variation of these amino acids directly affects the overall stiffness of the haemoglobin protein and has been selected by evolution to optimise its performance at each species’ own body temperature.
Dr. Andreas Stadler, Research Center Juelich in Germany: “We have shown a direct correlation between the resilience of these proteins and the average body temperature of the different species within which they operate. Haemoglobin acts as a highly sensitive molecular thermometer of body temperature. Our findings, which explain how evolution has optimised the vital job it carries out within different species, could prove particularly interesting to biological, bioengineering and biomedical research on red blood cells.”
Associate Professor Chris Garvey, a co-author of the paper: "In this work Australia's unique fauna, particularly monotremes and crocodylians, provides a unique perspective on evolutionary and physiological adaptation in haemoglobin. Including these more unusual members of the vertebrate family gave an insight into the adaptation of this protein to perform its vital role beyond the simple novelty of the exotic haemoglobin."
Prof Helmut Schober, Science Director at the Institut Laue-Langevin: “Neutrons are an increasingly important tool in a wide range of biological studies. The neutron spectroscopic technique used by Dr Stadler and his colleagues has been used to resolve the dynamic behaviour of a variety of functional biological systems, such as proteins and nucleic acids, from various organisms, membranes made up of lipids and protein, and even the entire proteome in living cells. New variations of the technique are continually being developed at institutes like the ILL to adapt to increasingly complex biological systems.”
More information: rsif.royalsocietypublishing.or … /rsif.2012.0364.full
Journal information: Journal of the Royal Society Interface
Provided by Institut Laue-Langevin










.jpg)