Ordered planar polymers created for the first time
(PhysOrg.com) -- Scientists under the direction of ETH Zurich have created a minor sensation in synthetic chemistry. They succeeded for the first time in producing regularly ordered planar polymers that form a kind of "molecular carpet" on a nanometre scale.
At ETH Zurich in 1920, the chemist Hermann Staudinger postulated the existence of macromolecules consisting of many identical modules strung together like a chain. For this he was initially rewarded with mockery and incomprehension in professional circles. But Staudinger was to be proved right: today the macromolecules described as polymers are known as plastics, and by 1950 one kilogram of them was already being produced per capita worldwide.
Today, more than ninety years after Staudinger’s discovery – for which the chemist was honoured with the Nobel Prize in 1953 – about 150 million tons of plastics are manufactured every year. A gigantic industry developed, without whose products our daily life is no longer imaginable.
A research group led by Professor A. Dieter Schlüter and Senior Lecturer Junji Sakamoto at the Polymers Institute of ETH Zurich has now succeeded in making a decisive breakthrough in the synthetic chemistry of polymers: they have created two-dimensional polymers for the first time.
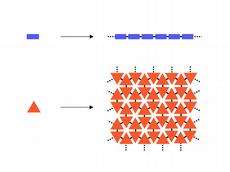
Polymers are formed when small single molecules known as monomers join together by chemical reactions like the links of a chain to form high molecular weight substances. Since qualifying as a lecturer, Schlüter was already occupied by the question of whether polymers can only polymerise linearly. Although graphene counts as a natural representative of a two-dimensional polymer – the carbon atoms in graphene form a honeycomb-like pattern through triple bonds – it cannot be synthesised in a controlled way.

Nevertheless, he said, if it is possible to produce giant molecules “one-dimensionally” from monomers, or for example molecules in pharmacology that are so small that they are practically “zero-dimensional”, why then should it not be possible to develop a synthetic chemistry that generates two-dimensional molecules? When Schlüter and Sakamoto met at ETH Zurich a few years ago, they discussed this topic intensively and together they looked for answers.
The crux of the matter was to create oligofunctional monomers in such a way that they join together purely two-dimensionally instead of linearly or even three-dimensionally. Polymers of this kind must have three or more covalent bonds between the regularly repeating units. The scientists had to find out which bonding chemistry and environment was most suitable for producing this kind of “molecular carpet”.
After intensive analyses of previous studies and the possible ways of generating two-dimensional polymers synthetically, they considered the synthesis at a water-air interface or in a single crystal, i.e. a crystal with a homogeneous layer lattice.
The researchers decided in favour of the second alternative: the doctoral student Patrick Kissel successfully used this to crystallise special monomers which he had prepared into layered hexagonal single crystals. For this he generated photochemically sensitive molecules for which such an arrangement is energetically optimum. When irradiated with light with a wavelength of 470 nanometres, the monomers polymerised in all the layers.
After this the researchers boiled the crystal in a suitable solvent to separate the individual layers from one another. Each layer represents a two-dimensional polymer. The fact that the team really had succeeded in producing sheet-like polymers with regular structures was shown by special studies in an electron microscope carried out by Empa researcher Rolf Erni and Marta Rossell from ETH Zurich at the Empa (Swiss Federal Laboratories for Materials Science and Technology).
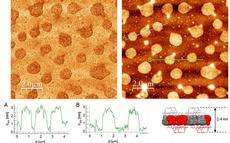
The polymerisation method that was developed is so gentle that all the monomer’s functional groups are also preserved at defined positions in the polymer. The researchers have complete structural control over the monomers in a way that would never be possible with graphene, for example, because that process would need to be carried out at enormously high temperatures. Sakamoto says, “Our synthetically manufactured polymers are not conductive like graphene, but on the other hand we would be able to use them for example to filter the tiniest molecules.”
In fact there are small defined holes with a diameter in the sub-nanometre range in the regularly arranged polymers. Moreover, tiny hexagons in the polymers, formed by benzene rings with three ester groups, can be removed by a simple hydrolytic process. This would form a “sieve” with an ordered structure suitable for the selective filtration of molecules.
However, before the researchers can think about practical applications, the task now is to characterise the material’s properties. According to Schlüter, this is mainly a job for the physicists. One of the exciting questions in this respect will be how a two-dimensional polymer behaves compared to a linear polymer, for which a good physical and technological understanding is available. Schlüter assumes that two-dimensional polymers could have a different physics and will therefore also find different applications.
He mentions the property of “elasticity” as an example: intertwined linear polymers enable a stretched rubber band to snap back as soon as it is released. But because flat sheets can hardly entangle together, this would probably not work with planar polymers. However, the researchers must first of all find a way to produce larger amounts and even larger sheet sizes.
The size of the crystals is currently only 50 micrometres. Sakamoto stresses that “those, however, are already enormous degrees of polymerisation at a molecular level.”
More information: Kissel P, Erni R, Schweizer WB, Rossell MD, King BT, Bauer T, Götzinger S, Schlüter AD & Sakamoto J: A two-dimensional polymer prepared by organic synthesis, Nature Chemistry (2012), advanced online publication, doi:10.1038/nchem.1265
Journal information: Nature Chemistry
Provided by ETH Zurich